In the field of metabolic disease therapy, Tirzepatide, as the world's first glucose‑dependent insulinotropic polypeptide (GIP)/glucagon‑like peptide‑1 (GLP‑1) dual‑target receptor agonist, has become one of the core medications for the treatment of type 2 diabetes, obesity and related metabolic complications due to its breakthrough therapeutic mechanism and remarkable clinical efficacy.
With its precisely optimized molecular structure, it features both long‑acting properties and dual agonistic activity. Since its approval by the U.S. FDA in 2022, it has rapidly penetrated major global medical markets, offering a novel therapeutic option for patients with metabolic disorders and driving the refined development of related pharmaceutical trade and supply chain services.
|
|
|
|
|
|
|
|
|
|
Full Record of Tirzepatide Procurement Process for the Spanish Client
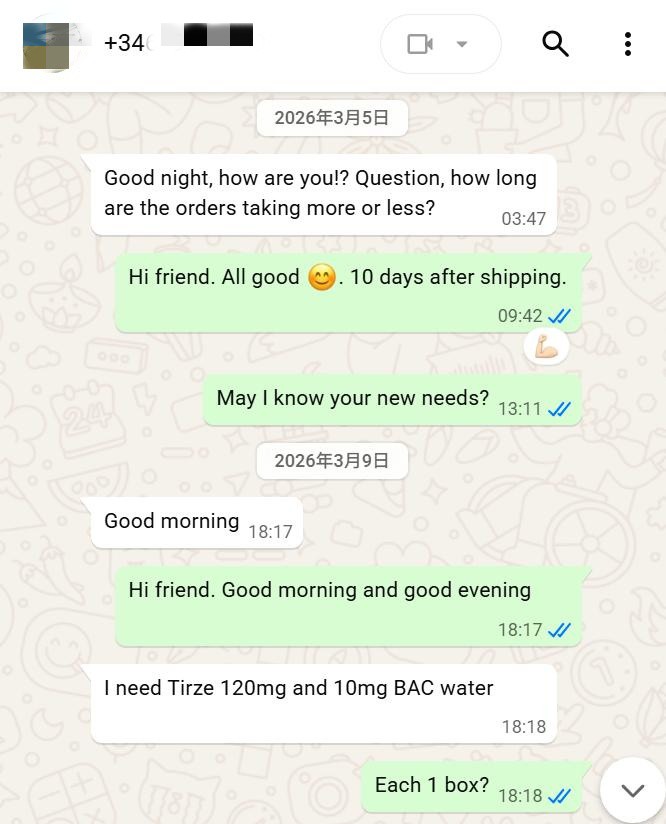
The client in this cooperation is a seasoned pharmaceutical procurement partner from Spain, who has long focused on the supply of internationally advanced raw materials for metabolic drugs. This procurement centered on 120mg Tirzepatide(https://en.wikipedia.org/wiki/Tirzepatide) and supporting 10ml BAC water, representing a targeted collaboration built on long‑term mutual trust.
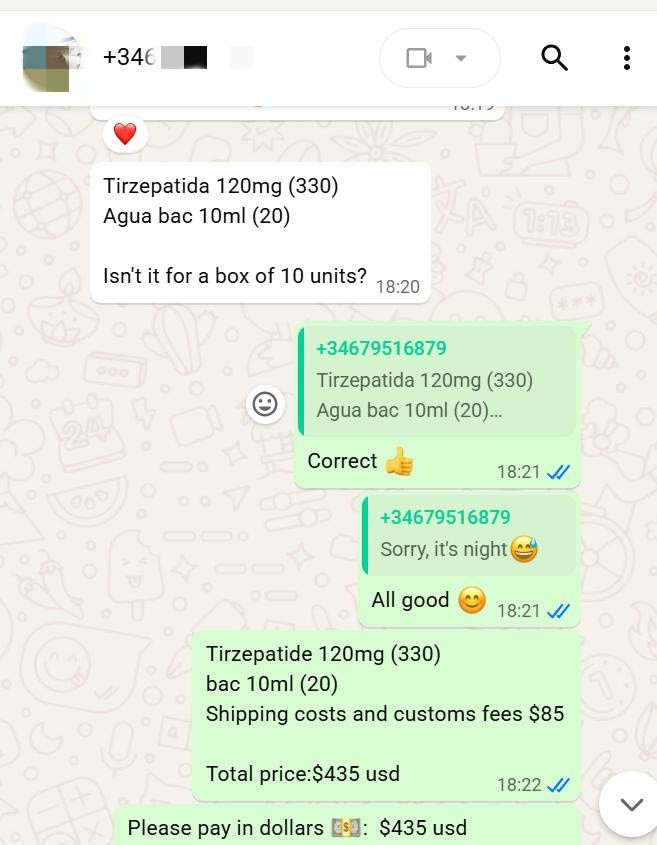
At the initial stage of the transaction, the client clearly specified procurement requirements via international communication channels, confirming the product specifications as 120mg Tirzepatide (USD 330) and 10ml BAC water (USD 20), and reached a consensus on core details including packaging specifications and delivery timelines.
In the cost accounting phase, based on international logistics standards and cross‑border customs clearance norms, we clearly listed freight and customs clearance fees at USD 85, resulting in a total quotation of USD 435, with full transparency and no hidden additional charges.
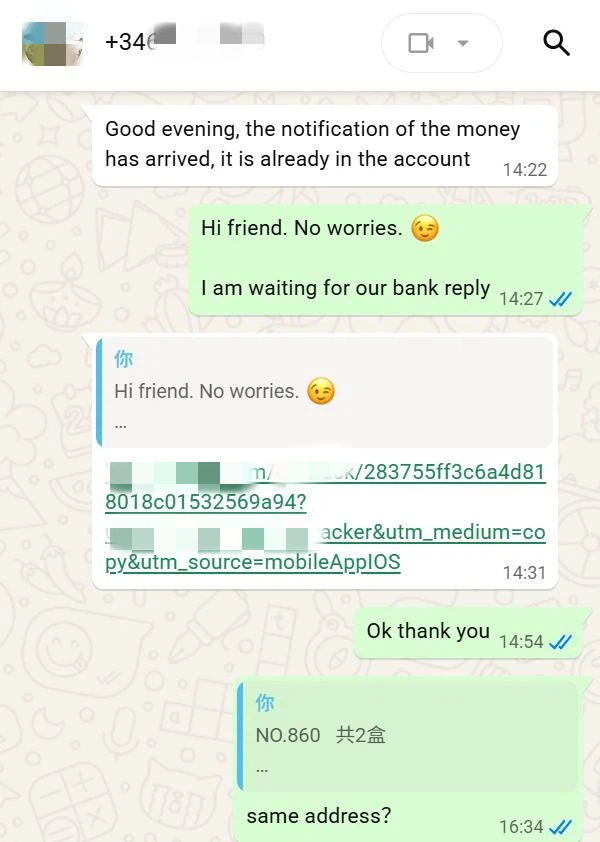
After confirming the quotation, the client completed full payment via the Wise platform efficiently, and we simultaneously confirmed receipt and filed the order.
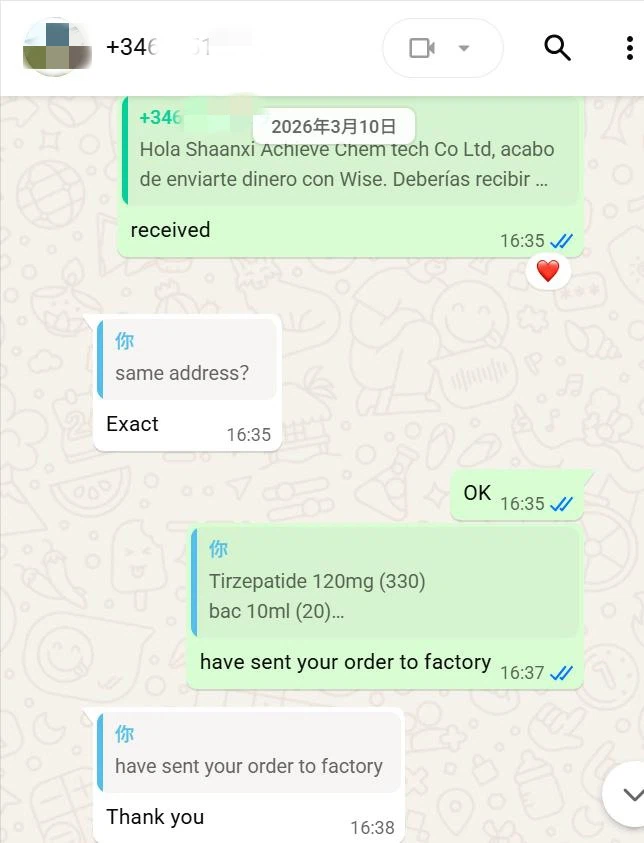
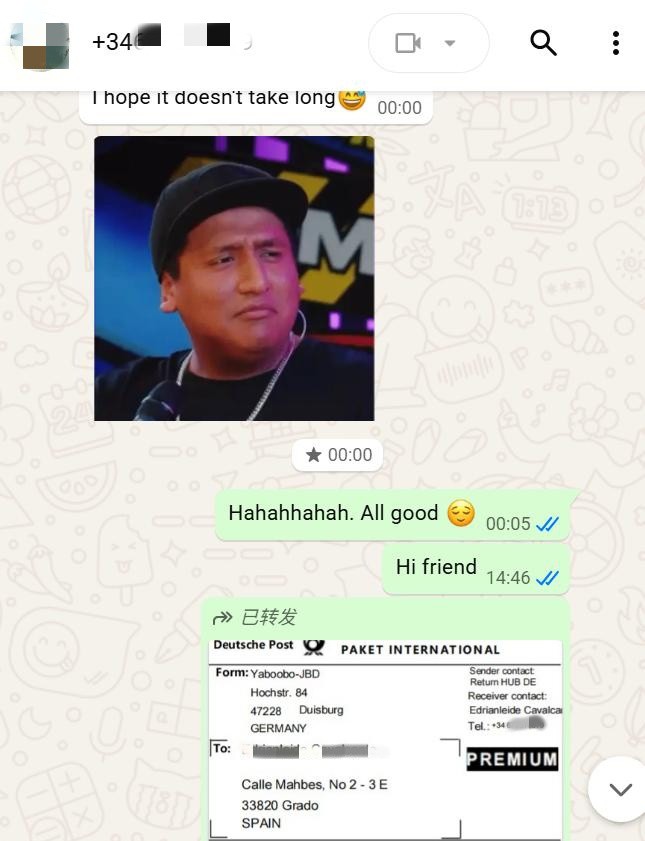
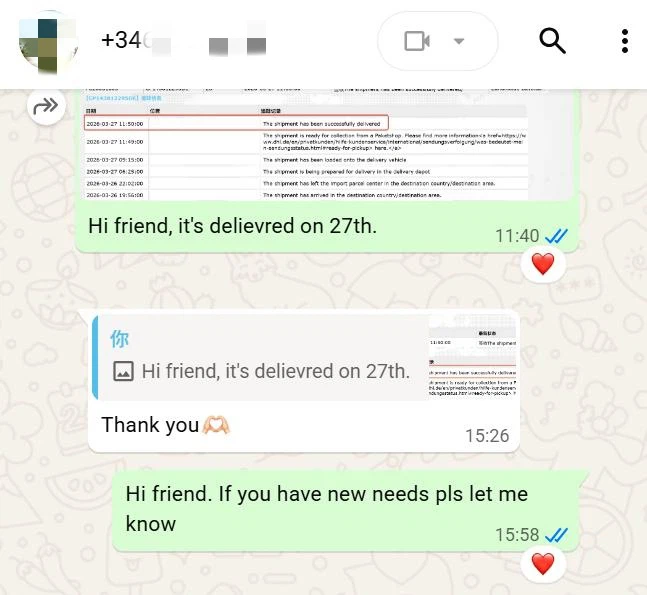
During order execution, we promptly initiated stock preparation, finished product packaging in strict accordance with quality control standards for pharmaceutical raw materials, verified consistency between the shipping address and the client's registered address, and arranged international delivery via Deutsche Post, providing a real‑time logistics tracking number to synchronize cargo status.
Ultimately, the goods successfully cleared cross‑border customs and were accurately delivered to the designated address in Spain.
The entire process, from order confirmation to final delivery, was seamlessly and efficiently coordinated, realizing precise, safe and high‑efficiency cross‑border supply of pharmaceutical raw materials.
Core Application Scenarios of Tirzepatide
1.1 Core Therapeutic Agent for Type 2 Diabetes
As a highly prevalent chronic metabolic disease worldwide, type 2 diabetes has long relied on traditional regimens focused on single‑target glycemic control, which can hardly balance metabolic homeostasis and complication prevention. With its dual‑target activation mechanism, Tirzepatide has become a preferred option for type 2 diabetes, especially for adult patients with inadequate glycemic control following treatment with metformin or sulfonylureas, and can also be combined with insulin for optimized therapy.
1.2 Long‑Term Weight Management for Obese and Overweight Individuals
The global population of obese individuals continues to expand; obesity not only impairs quality of life but also serves as a key risk factor for multiple chronic diseases including type 2 diabetes and cardiovascular diseases. Tirzepatide is approved for long‑term weight management in adults with obesity (BMI ≥ 28 kg/m²) or overweight (BMI ≥ 24 kg/m²) complicated by weight‑related comorbidities, with weight‑loss effects significantly superior to similar single‑target agents.
1.3 Adjunctive Therapy for Metabolic Complications
Beyond its core indications, Tirzepatide demonstrates significant value in treating metabolic complications. In 2024, it was approved for the management of moderate to severe obstructive sleep apnea (OSA) in obese patients. By improving metabolic status and reducing fat deposition around the airways, it effectively relieves apneic symptoms and enhances sleep quality.
Furthermore, in cardiovascular protection, Tirzepatide reduces the risk of cardiovascular death or heart failure worsening by 38% in patients with heart failure with preserved ejection fraction (HFpEF). It also improves lipid profiles, lowering triglycerides by 18%–31% and low‑density lipoprotein cholesterol by 5%–15%, and reducing inflammatory cytokine release, providing a new comprehensive therapeutic option for patients with metabolic diseases complicated by cardiovascular disorders and expanding its clinical applications.
Development Prospects and Industrial Value of Tirzepatide
3.1 Continuous Expansion of Clinical Applications
With advancing clinical research, the application scope of Tirzepatide is broadening steadily. Studies on its use in non‑alcoholic steatohepatitis (NASH) and metabolic dysfunction‑associated steatohepatitis (MASH) have entered critical stages. Animal experiments confirm its ability to reduce hepatic fat accumulation and improve fibrosis markers, positioning it as a potential breakthrough drug for NASH.
Meanwhile, intervention studies in prediabetic populations show that Tirzepatide improves insulin resistance and delays progression to overt type 2 diabetes, shifting metabolic disease management from late treatment toward early intervention. Its potential applications in polycystic ovary syndrome, Alzheimer's disease and other fields are also under exploration, and it is expected to become a core drug for metabolic disorders across multiple indications.
3.2 Prominent Market Scale and Industrial Value
The growing global prevalence of obesity and type 2 diabetes has created vast market potential for Tirzepatide. In 2025, driven by its outstanding weight‑loss and glucose‑lowering efficacy, it became one of the world's top‑selling blockbuster pharmaceuticals, with market coverage extending from Europe and the Americas to emerging markets in Asia and Africa. In China, Tirzepatide has been approved for inclusion in the national medical insurance catalogue, significantly reducing patient costs and promoting wider access.
From a pharmaceutical trade perspective, the cross‑border supply of Tirzepatide imposes stringent requirements on supply chains, with every stage - from raw material production and quality control testing to international logistics and customs clearance - strictly complying with pharmaceutical industry standards. This procurement cooperation with the Spanish client serves as a typical example of refined cross‑border supply chain operations for pharmaceutical raw materials, reflecting strong market demand for Tirzepatide and highlighting the core role of professional pharmaceutical supply chain services in ensuring global drug availability.
3.3 Promotion of Industrial Technology and R&D
The successful development and application of Tirzepatide have advanced industrial progress in peptide drug synthesis, long‑acting formulation development, and dual‑target drug design. Its molecular modification techniques provide a reference for the research of similar metabolic drugs, accelerating the development of novel agents such as GIP/GLP‑1 dual agonists and GLP‑1/glucagon dual agonists.
Meanwhile, accumulated clinical data have provided important evidence for updating metabolic disease treatment guidelines, driving a global shift from single‑target glycemic control to comprehensive metabolic management. In the future, breakthroughs in gene editing, peptide synthesis and related technologies are expected to further optimize production costs and improve drug accessibility, benefiting more patients with metabolic diseases worldwide.
Conclusion
From precise procurement to efficient delivery for the Spanish client, this cooperation represents not only a practical case of cross‑border pharmaceutical raw material trade but also a microcosm of the refined operation of Tirzepatide's global supply system.As a breakthrough drug in metabolic disease therapy, Tirzepatide, with its unique dual‑target mechanism, wide application scenarios and remarkable clinical efficacy, is continuously reshaping the landscape of metabolic disease treatment.
Professional pharmaceutical supply chain services, acting as a bridge between drug R&D and clinical application, provide precise, standardized and efficient operations that serve as the core support for ensuring the global supply of blockbuster drugs such as Tirzepatide.Moving forward, with deeper clinical research and upgraded supply chain technologies, Tirzepatide will further expand its application boundaries, drive deeper integration between pharmaceutical trade and industrial development, and deliver greater health benefits to patients with metabolic diseases worldwide.