Skeletal health, as an important foundation for the stable operation of human functions, runs through the entire process of human life activities and directly affects the physical activity ability and quality of life. When there is an imbalance in bone metabolism and insufficient nutritional supply, there will be persistent abnormal states such as fragility and vulnerability. These states not only limit daily activities, but also may cause various types of fracture hazards, thereby increasing physical burden and affecting self-care ability. At this time, it is urgent to use scientific and targeted intervention methods to alleviate bone fragility and reduce health risks.
Teriparatide Acetate, as a targeted intervention substance, has shown particularly significant adaptation value in two specific bone care scenarios. These scenarios correspond to the severe bone fragility problem faced by postmenopausal women and the bone loss problem caused by drug effects in long-term estrogen users. For these two scenarios, the application logic of it, the core characteristics of the adapted population, and the protective effects in practical applications can be interpreted in detail from multiple dimensions and levels, providing scientific and practical reference for the bone health protection of relevant populations and helping them better cope with bony tissue fragility hazards and maintain physical health.
 |
 |



Teriparatide Acetate COA
 |
||
| Certificate of Analysis | ||
| Compound name | Teriparatide Acetate | |
| Grade | Pharmaceutical grade | |
| CAS No. | 52232-67-4 | |
| Quantity | 38g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090086 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.54% |
| Loss on drying | ≤1.0% | 0.42% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.98% |
| Single impurity | <0.8% | 0.52% |
| Total microbial count | ≤750cfu/g | 95 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 500ppm |
| Storage | Store in a sealed, dark, and dry place below -20°C | |
|
|
||
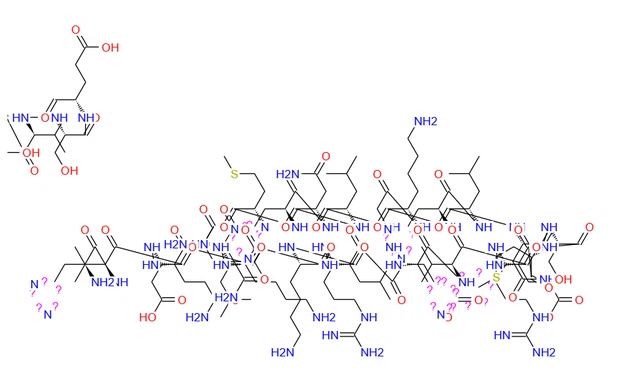
| Chemical Formula | C181H291N55O51S2 |
| Exact Mass | 4115.13 |
| Molecular Weight | 4117.77 |
| m/z | z: 4116.13 (100.0%), 4117.14 (97.3%), 4115.13 (51.1%), 4118.14 (36.1%), 4119.14 (29.7%), 4118.14 (26.6%), 4117.13 (20.3%), 4118.13 (19.8%), 4118.14 (10.5%), 4116.13 (10.4%), 4119.14 (10.2%), 4118.13 (9.0%), 4119.13 (8.8%), 4119.14 (7.3%), 4120.15 (7.1%), 4120.14 (5.7%), 4119.14 (5.4%), 4117.14 (5.4%), 4117.13 (4.6%), 4120.14 (4.1%), 4120.15 (3.8%), 4120.15 (3.1%), 4120.15 (2.8%), 4119.13 (1.8%), 4121.14 (1.8%), 4120.13 (1.8%), 4118.14 (1.6%), 4118.13 (1.4%), 4119.13 (1.3%), 4121.13 (1.2%), 4121.15 (1.1%) |
| Elemental Analysis | C, 52.80; H, 7.12; N, 18.71; O, 19.82; S, 1.56 |

Adaptation application for severe skeletal fragility in postmenopausal women
The application of this substance in severe bony tissue fragility in postmenopausal women focuses on the group with higher risk of fractures, and through targeted intervention, reduces the possibility of fractures in different parts of the body. Its adaptation logic and practical effects can be developed from the following points:

The core definition of the suitable population mainly refers to the female population with significantly decreased bony tissue stability and hidden risk of fractures after menopause. This group of people often have the characteristics of a long menopausal period and a decline in their own bony tissue maintenance ability. Some of them are also accompanied by low body weight, insufficient daily activity, or bony tissue related family risks. The toughness and resistance of their bony tissue are significantly lower than those of ordinary postmenopausal women, making them a high-risk group for fractures and the core target of this substance.
The prevention and control value of vertebral fractures mainly lies in alleviating the fragile state of vertebral bony tissue and reducing vertebral injuries caused by minor external forces. Postmenopausal women are prone to problems such as nutrient loss and loose structure in their vertebral bony tissue . Slight activities such as bending and turning around in daily life may cause vertebral fractures. However, this substance can gradually improve the stability of vertebral bony tissue by adapting to the metabolic characteristics of this population, reducing the probability of vertebral fractures and alleviating the physical discomfort and inconvenience caused by fractures.

The prevention and control significance of non vertebral fractures focuses on non vertebral bony tissue parts such as limbs and ribs, and reduces the probability of fractures in these areas by enhancing the toughness of the bony tissue themselves. Non vertebral fractures are often caused by accidental bumps, minor falls, and other situations. However, postmenopausal women with severe bony tissue fragility have a significantly increased risk of such fractures. This substance can specifically enhance the anti damage ability of non vertebral bony tissue, reduce the occurrence of fractures in unexpected situations, and provide more comprehensive bony tissue protection for their daily activities.
Data source:
Compston J. Glucocorticoid-induced osteoporosis: an update[J]. Endocrine, 2018, 61(1): 7-16.
Taylor AD. Anabolics in the management of glucocorticoid-induced osteoporosis: an evidence-based review[J]. Journal of bony tissue and Mineral Research, 2019, 34(10): 1789-1802.
Adaptive application of hormone induced skeletal fragility
The population who use hormones for a long time is prone to sustained effects on their bony tissue health, leading to problems such as fragile bony tissue and increased risk of fractures. The adaptation and application of Teriparatide Acetate for such situations focuses on the core needs of the long-term hormone users, alleviating the bone damage caused by hormones. Specific details can be explained from the following points:

The core characteristics of the adapted population are mainly those who rely on long-term hormone regulation to control their own diseases and significantly increase bony tissue fragility. This group of people often require long-term use of hormones to control their condition due to autoimmune diseases, chronic inflammation, and other issues. The duration of medication usually reaches three months or more. Some people also have high hormone usage, which inhibits bone nutrient absorption and metabolism, and increases the risk of fractures compared to the general population. Therefore, targeted interventions should be carried out using this substance.
The adaptation logic of hormone induced bony tissue fragility is centered on fitting the characteristics of hormone effects on bony tissue and alleviating bony tissue loss. Long term use of hormones can quietly weaken the self-care ability of bony tissue, leading to gradual loss of bone mass, decreased bony tissue toughness, and even hidden bone damage. However, this substance can specifically counteract some of the negative effects of hormones, improve bony tissue metabolism, reduce bone loss, alleviate the progression of bone fragility, and provide bony tissue protection for people who use hormones for a long time.

The prevention and control effect of high risk of fractures is mainly aimed at the high incidence of fractures in the population who use hormones for a long time, reducing the possibility of various types of fractures. The risk of fractures in this group of people is not only reflected in common parts such as the vertebral body and limbs, but also in the overall fragility of the bones, which may cause fractures with slight external forces. This substance can enhance the stability and resistance of the bony tissue, reduce the risk of fractures in a targeted manner, and is particularly suitable for long-term hormone users with high fracture risk. It can help them control underlying diseases while protecting bony tissue health.
Data source:
Indian Journal of Endocrinology and Metabolism. Glucocorticoid-induced Osteoporosis[J]. 2017, 21(6): 652-658.
8. Dingxiangyuan New Drug Research Center Clinical Application Guidelines for Teriparatide Acetate [M]. Beijing: People's Health Publishing House, 2023
CIMA. FICHA TECNICA TERIPARATIDA TEVA[R]. 2025.

In summary, the clinical application of Teriparatide Acetate in the aforementioned two scenarios is specifically designed for individuals at high risk of osteoporotic fractures. By adaptively matching the bony tissue metabolic status, bony tissue mineral density level, and physiological characteristics of different patient groups, it can effectively slow down bony tissue mass loss, promote bony tissue formation, and significantly lower the incidence of fragility fractures, thereby offering reliable protection and clinical support for the skeletal health of these populations. To fully realize its application value, its use must be rationally combined with the individual conditions, physical baseline, and disease status of specific populations, so as to better reduce fracture risk, maintain stable bony tissue metabolism and skeletal function, and improve long-term quality of life.
References
Chinese Medical Association Osteoporosis and Bone Mineral Diseases Branch Guidelines for Diagnosis and Treatment of Postmenopausal Osteoporosis (2022 Edition) [J]. Chinese Journal of Osteoporosis and bony tissue Mineral Diseases, 2022, 15 (3): 233-254
Vithran DA, Essien AE, Rahmati M, et al. Teriparatide in postmenopausal osteoporosis: a systematic review and meta-analysis[J]. European Orthopaedics Reviews, 2024, 19(2): 1-10.
Orthopedics Department of Linyi Central Hospital Research on the Prevention and Control of Corticosteroid Related Osteoporosis [J]. Shandong Medical Journal, 2025, 65 (12): 78-82
FAQ
- What is the difference between teriparatide and teriparatide acetate?
Teriparatide is a recombinant human parathyroid hormone (PTH) administered as daily subcutaneous injections, whereas teriparatide acetate is a biologically active fragment containing the N-terminal 34 amino acids of human PTH administered as weekly subcutaneous injections.
- Can this acetate cause weight loss?
You shouldn't have weight gain or weight loss as a side effect of Forteo. People taking Forteo in clinical trials didn't report either of these side effects. However, weight loss can be a symptom of a high calcium level. This is a rare, serious side effect of Forteo.
Hot Tags: teriparatide acetate, China teriparatide acetate manufacturers, suppliers