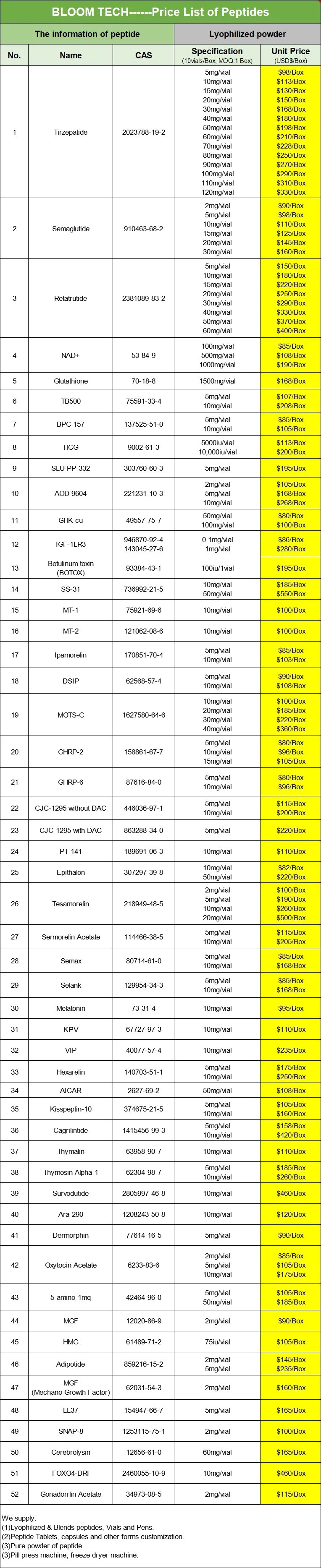
The PT 141 peptide spray is typically a nasal spray formulation, designed to achieve non-invasive drug delivery through the rich capillary network of the nasal mucosa. The core of this formulation is to dissolve PT-141 powder in a sterile, isotonic, and biocompatible solution, and add stabilizers and possible absorption enhancers, before being filled into a spray device with a quantitative valve. Users spray the drug into the nasal cavity, bypassing the first-pass effect and directly entering the systemic circulation. This theoretically provides better bioavailability than oral administration and a more convenient usage experience than injection. However, this formulation faces significant challenges: enzymatic degradation in the nasal environment, rapid loss due to mucosal cilia clearance, significant individual absorption differences, and common side effects such as nasal irritation, unpleasant odor, or temporary burning sensation. Its actual efficacy often fluctuates greatly due to differences in formulation technology. Most commercially available products lack strict clinical pharmacological validation, creating a gap between the convenience promise and reliable efficacy that consumers need to weigh for themselves.
Our Products Form
|
|
|
|
|
|
|
|

PT-141 COA


The instability of bioavailability
The bioavailability of PT 141 Peptide Spray (Bremer Langdan Peptide Nasal Spray) is influenced by multiple factors. Its instability is mainly manifested in aspects such as the mode of administration, individual differences, drug interactions, and environmental conditions. The following analysis is conducted from the core mechanism and influencing factors:
Limitations of Nasal Administration Method
PT-141 is administered in the form of nasal spray, and its bioavailability is directly influenced by the absorption efficiency of the nasal mucosa:
Mucosal barrier
The nasal mucosa is composed of multiple layers of cells. Drugs need to penetrate the mucus layer, epithelial cells, and capillary endothelium before entering the bloodstream. Although the molecular weight (approximately 1025 Da) and liposolubility of PT-141 facilitate penetration, the ciliary clearance function of the nasal mucosa may shorten the drug's residence time and reduce the absorption amount.
Local irritation
High concentrations of PT-141 may cause congestion or inflammation of the nasal mucosa, further hindering drug absorption. For example, some patients experience temporary nasal congestion or runny nose after use, which may reduce the drug's retention in the nasal cavity.
Differences in administration techniques: The particle size of the spray, the spray angle, and the patient's operation method (such as inhalation depth) all affect the distribution of the drug in the nasal cavity. If the spray does not evenly cover the absorption area (such as the middle and lower turbinates), the bioavailability may significantly decrease.
Impact of individual physiological differences
Different individuals' physiological characteristics may cause fluctuations in the bioavailability of PT-141:

Nasal cavity structure
Abnormalities in the nasal structure, such as deviation of the nasal septum, hypertrophy of the nasal conchae, or nasal cavity narrowing, may alter the flow path of drugs in the nasal cavity and affect the absorption efficiency.
Metabolic enzyme activity
Metabolic enzymes in the nasal mucosa (such as cytochrome P450) may locally metabolize PT-141, reducing the amount of drug entering the system. Enzyme activity is influenced by genetic polymorphisms and varies among different individuals.


Disease state
Patients with rhinitis, sinusitis, or allergic diseases may have abnormal nasal mucosa permeability, ciliary function, and inflammatory levels, leading to unstable absorption of PT-141. For example, the bioavailability of patients with chronic rhinitis may be reduced by 30%-50% compared to healthy individuals.
Risk of drug interactions
The combination of PT-141 with other drugs may affect its bioavailability through competitive absorption or metabolic interference:
Local nasal medication
Concurrent use of decongestants (such as pseudoephedrine) or antihistamines (such as cetirizine) may alter the state of the nasal mucosa and affect the permeability of PT-141. For instance, decongestants reduce mucosal blood flow by constricting blood vessels, which may slow down the drug absorption rate.


Systemic medication
P450 enzyme inducers (such as rifampicin) or inhibitors (such as ketoconazole) may indirectly affect the clearance rate of PT-141 by regulating the activity of metabolic enzymes. Although the contribution of nasal local metabolism is not yet clear, the theoretical risk exists.
Constraints from Environment and Storage Conditions
The stability of PT-141 is affected by environmental factors such as temperature, humidity, and light:
Temperature sensitivity
The PT-141 powder is stable at room temperature, but the solution after reconstitution needs to be stored refrigerated (2-8℃). If the storage temperature is too high, the drug may degrade, resulting in a dosage lower than the labeled value.
Light exposure and oxidation
Exposure to ultraviolet light or oxygen may cause changes in the molecular structure of PT-141, reducing the content of the active ingredient. For example, solutions not stored in the dark may become ineffective within a few weeks.
Reconstitution procedure
If patients reconstitute the drug by themselves and do not strictly follow the instructions (such as using the wrong solvent or not mixing well enough), it may lead to uneven drug concentration, further exacerbating fluctuations in bioavailability.
Clinical response strategies
To improve the stability of the bioavailability of PT-141, the following measures can be taken:
Optimize drug administration techniques
Train patients on how to use the spray device correctly (such as inhaling deeply before spraying the medicine) to ensure uniform distribution of the drug.
Individualized dosage adjustment
Adjust the dosage based on the patient's nasal condition (such as whether they have nasal inflammation) and metabolic characteristics, and make dynamic adjustments as needed.
Avoid drug interactions
When using other drugs concurrently, assess the impact on the absorption or metabolism of PT-141, and adjust the medication time or regimen if necessary.
Strict storage conditions
The reconstituted solution should be refrigerated and used within 30 days. Avoid repeated freezing and thawing or exposure to light.
Monotherapy: Exploring the Unique Value of Central Regulation
PT 141 Peptide Spray (Bremerwanda Peptide Nasal Spray), as a melanocortin receptor (MC) agonist, has demonstrated unique central regulatory value in the field of male erectile dysfunction (ED) treatment. It activates receptors such as MC4R in the central nervous system to regulate the release of neurotransmitters, thereby improving sexual function and providing a new approach for ED treatment.
|
|
|
|
Central regulatory mechanism: Dopamine as the bridge between sex drive and the brain
The core mechanism of PT 141 lies in its specific binding to the MC4R receptor. MC4R is widely distributed in the central nervous system, especially in regions closely related to sexual behavior regulation, such as the hypothalamus and the limbic system. After activating the MC4R, PT 141 can promote the release of neurotransmitters such as dopamine, norepinephrine, and oxytocin. Dopamine is known as the "happiness hormone", and its elevated level can directly stimulate sexual desire and enhance sexual motivation; norepinephrine enhances sexual arousal by promoting vascular contraction and neural excitation; oxytocin is involved in the formation of emotional connection and sexual satisfaction. This multi-dimensional regulatory mechanism enables PT 141 to improve the sexual function of patients with ED at the central level, rather than relying solely on peripheral vasodilation.
Clinical evidence: Exploration from women to men
The central regulatory value of PT 141 peptide spray has been verified by multiple clinical trials. In the treatment of female sexual desire disorder (HSDD), PT 141 has been approved by the FDA and has become the first non-hormonal therapeutic drug. Its phase III clinical trial included 1,267 pre-menopausal women, and the results showed that patients in the 1.75mg dose group had a significant improvement in sexual desire scores, and the safety was good. Common adverse reactions were mild nausea, flushing, and headache. This success laid the foundation for the application of PT 141 in male ED treatment.
In the treatment of male erectile dysfunction (ED), the single-agent exploration of PT 141 has also made progress. Although early studies were limited due to the issue of elevated blood pressure, subsequent research gradually overcame this obstacle by optimizing the administration method (such as nasal spray) and adjusting the dosage. For instance, a trial for patients with ineffective sildenafil-induced ED showed that the PT 141 nasal spray could significantly improve erectile function, and the blood pressure fluctuations were within a controllable range. This indicates that the mechanism by which PT 141 achieves sexual function improvement through central regulation is also effective in male patients and has unique advantages.
Unique Value: Breaking Through the Limitations of Traditional Treatments
The central regulatory value of PT 141 is manifested in several aspects. Firstly, its mechanism of action is completely different from that of traditional ED drugs (such as PDE5 inhibitors). PDE5 inhibitors improve erectile function by dilating peripheral blood vessels, but have limited effects on central ED (such as ED caused by psychological factors or nerve damage); while PT 141 directly acts on the central nervous system, it can improve the sexual motivation and arousal ability of patients with psychogenic ED. Secondly, PT 141 does not rely on vasodilation, so it is safer for ED patients with cardiovascular diseases. For example, a trial on patients with hypertension and ED showed that PT 141 did not significantly affect blood pressure, while PDE5 inhibitors may increase the risk of hypotension. Finally, the rapid onset (about 45 minutes) and long-lasting maintenance (6-8 hours) characteristics of PT 141 make it more in line with the immediate needs of sexual activities.
Future directions: Optimizing administration and expanding indications
Although PT 141 shows potential in central regulation of ED treatment, its clinical application still requires further optimization. For instance, the bioavailability of nasal spray may be affected by the state of the nasal mucosa. In the future, this can be improved through nanotechnology or liposome carriers to enhance drug permeability. Additionally, the indications of PT 141 can be expanded to other areas of sexual dysfunction, such as premature ejaculation (PE) or sexual anxiety. Through combination therapy or personalized dose adjustment, a more comprehensive sexual health management can be achieved.
PT 141 Peptide Spray offers unique value in the treatment of erectile dysfunction through a central regulatory mechanism. Its dopamine-mediated enhancement of sexual desire, its cardiovascular safety profile, and its rapid and long-lasting effects make it an important supplement to traditional ED medications. With the optimization of the administration method and the expansion of indications, PT 141 is expected to play a greater role in the field of sexual medicine in the future.
|
|
|
|
FAQ
1. Is it really effective?
The effect varies from person to person and is highly dependent on the formulation technology. Due to its unstable absorption (easily cleared and degraded by the nasal cavity), the actual effect may be weaker than that of the injection, and the onset time and intensity fluctuate greatly. Many commercially available sprays lack rigorous clinical data support.
2. How to Use?
Usually, sit upright and insert the nozzle into one nostril. While inhaling, press down on the nozzle. After using it, slightly tilt your head back for a moment. Make sure to follow the dosage instructions provided by the product (usually the single spray volume), and never increase the frequency or dosage without permission to avoid exacerbating side effects.
3. What are the main drawbacks?
The key drawbacks include: nasal irritation or discomfort, less reliable effectiveness compared to injections, generally lower cost-effectiveness (due to absorption efficiency issues, the cost of the effective dose is higher), and the uneven quality of market products, with the risk of inaccurate purity and concentration.
Hot Tags: pt 141 peptide spray, China pt 141 peptide spray manufacturers, suppliers, CJC 1295 NO DAC 2mg, IGF 1 LR3 Spray, IGF 1 LR3 Tablet, Ipamorelin Injection, PT 141 Injection, Sermorelin Capsules

























