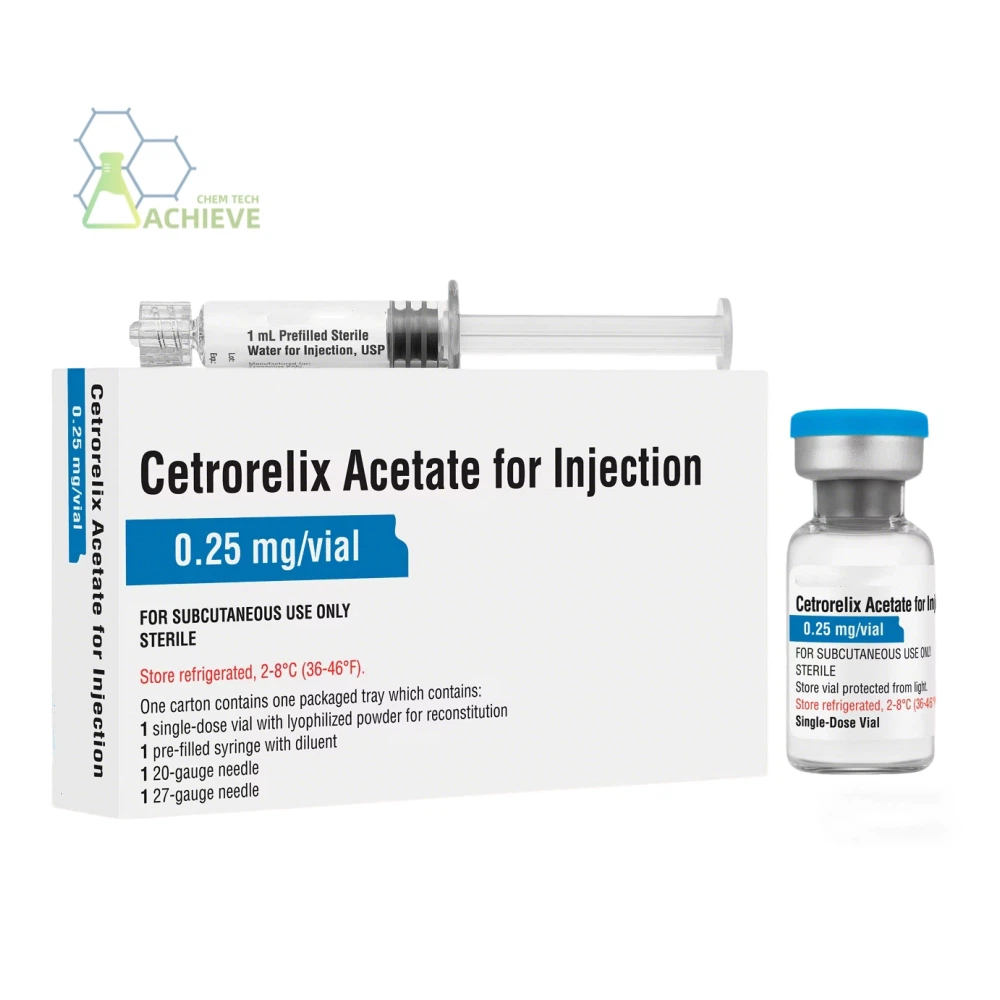
Cetrorelix acetate injection is a third-generation GnRH antagonist injection, primarily indicated for controlled ovarian stimulation in assisted reproductive technology. It precisely prevents premature ovulation, thereby improving oocyte retrieval and in vitro fertilization success rates. Key features include rapid onset of action within 1 hour and sustained, stable control of luteinizing hormone (LH). It requires no long-term priming phase, enables flexible initiation of antagonist protocols, and significantly shortens treatment duration.
Our Products Form




Cetrorelix acetate COA
 |
||
| Certificate of Analysis | ||
| Compound name | Cetrorelix acetate | |
| Grade | Pharmaceutical grade | |
| CAS No. | 145672-81-7 | |
| Quantity | 52g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090056 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.45% |
| Loss on drying | ≤1.0% | 0.38% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.90% |
| Single impurity | <0.8% | 0.46% |
| Total microbial count | ≤750cfu/g | 428 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 516ppm |
| Storage | Store in a sealed, dark, and dry place below -20°C | |
|
|
||
|
|
||
| Chemical Formula | C70H92ClN17O14.C2H4O2 |
| Exact Mass | 1489.69 |
| Molecular Weight | 1491.11 |
| m/z | 1489.69(100.0%), 1490.69(77.9%), 1491.69(32.0%), 1431.68(28.3%), 1432.67(24.2%), 1433.67(9.0%), 1430.67(6.3%), 1432.68(6.1%), 1431.67(4.5%), 1431.67(2.9%), 1434.68(2.2%), 1432.68(2.2%), 1432.66(1.9%), 1432.67(1.8%), 1433.67(1.5%), 1433.68(1.3%), 1430.68(1.1%) |
| Elemental Analysis | C,58.00; H,6.49; C,2.38; N,15.97; O,17.17 |

Fundamental Mechanism of Action
The use of cetrorelix acetate injection in assisted reproductive technology relies on its unique pharmacological properties to maintain a stable hormonal environment during controlled ovarian stimulation. Its mechanism is as follows:
The drug competitively binds to GnRH receptors on pituitary cell membranes, completely blocking the binding of endogenous GnRH to its receptors, thereby directly inhibiting the secretion of LH and follicle-stimulating hormone (FSH) without an initial stimulatory effect. Unlike GnRH agonists, it does not undergo a "stimulation-then-suppression" phase; hormonal inhibition is achieved immediately after administration, preventing abnormal follicular development caused by transient elevations in LH levels.
Its inhibitory effect is clearly dose-dependent: daily administration of 0.25 mg maintains stable suppression for 24 hours, while a single 3 mg dose sustains suppression for at least 4 days, with approximately 70% inhibition of LH still observed on day 4. Following discontinuation, pituitary function recovers rapidly, and endogenous LH and FSH secretion return to normal without impairing subsequent follicular development or spontaneous ovulation. This profile of "rapid suppression, steady regulation, and reversible recovery" aligns perfectly with the need for precise hormonal control in assisted reproduction, providing a solid pharmacological basis for its core clinical applications.
Information source: EMD Serono Cetrotide® 0.25 mg Prescribing Information; China Medical Information Platform Pharmacological Action of Cetrorelix Acetate for Injection; Electronic Medicines Compendium (EMC) Pharmacological Mechanism of Cetrorelix Acetate.
Core Clinical Indications
2.1 Prevention of Premature Ovulation in Controlled Ovarian Stimulation (COS)
Controlled ovarian stimulation is a central step in assisted reproductive techniques such as IVF-ET and ICSI. Its purpose is to stimulate the ovaries with exogenous gonadotropins (e.g., FSH, hMG) to promote synchronous maturation of multiple follicles, yielding sufficient high-quality oocytes for subsequent fertilization, embryo culture, and transfer.
However, during gonadotropin stimulation, accelerated follicular development often leads to a premature LH surge – an early rise in LH levels that triggers premature ovulation of immature oocytes, resulting in failed oocyte retrieval, reduced oocyte quality, and severely compromised ART success.A primary indication of cetrorelix acetate injection is the precise suppression of premature LH surges to prevent premature ovulation.
By maintaining consistent inhibition of pituitary LH secretion, it allows follicles to continue developing under gonadotropin support until optimal maturity (typically 18–20 mm in diameter).


In clinical practice, timing of cetrorelix administration is guided by follicular monitoring (ultrasound assessment of follicle size and number combined with hormonal measurements), ensuring synchronized follicular maturation.Human chorionic gonadotropin (hCG) is then administered at the optimal window to trigger ovulation and improve oocyte retrieval rates.
Clinical data show that use of cetrorelix during COS reduces the incidence of premature LH surges to below 5%, significantly lowering the risk of retrieval failure and increasing the proportion of high-quality oocytes by 12%–18% compared with untreated controls. It also improves outcomes in patients with unstable ovarian response or a history of premature LH surges, reducing cycle cancellations due to ovulatory disorders.
Information source: Journal of Reproductive Medicine, 2025, Application of Domestic Cetrorelix in GnRH Antagonist Protocols; Medicover Hospitals Clinical Practice Guidelines for Cetrorelix.
Ovarian hyperstimulation syndrome (OHSS) is a serious complication of controlled ovarian stimulation, with an overall incidence of 5%–10% and rates exceeding 20% in high-risk populations (e.g., polycystic ovary syndrome, high ovarian responders, young patients). Pathogenically, excessive ovarian stimulation by exogenous gonadotropins leads to marked ovarian enlargement and increased vascular permeability, resulting in ascites, pleural effusion, electrolyte imbalance, thrombosis, and potentially life-threatening conditions. Premature LH surges and ovarian hyperresponse induced by exogenous hCG are major triggers of OHSS.
It effectively reduces OHSS risk through dual mechanisms. First, it suppresses LH surges, diminishing ovarian hyperresponse to gonadotropins and preventing excessive follicle numbers and supraphysiological estradiol levels – a key driver of increased vascular permeability in OHSS. Second, it inhibits excessive luteinization of the ovaries, reducing the release of vasoactive substances such as vascular endothelial growth factor, thereby lowering vascular permeability and decreasing ascites and pleural effusion formation.
Clinical studies confirm that antagonist protocols using cetrorelix reduce overall OHSS incidence by 40%–50% and moderate-to-severe OHSS by more than 60% compared with conventional long GnRH agonist protocols. In high responders including PCOS patients, those with prior OHSS, and good ovarian reserve, cetrorelix improves treatment safety, avoids cycle cancellation due to complications, and reduces patient discomfort and medical costs. Its reversibility allows OHSS prevention without compromising subsequent follicular maturation or embryo transfer, achieving both efficacy and safety.
Information source: Chinese Journal of Reproduction and Contraception, 2024, Advances in Clinical Research on GnRH Antagonists in OHSS Prevention; China Medical Information Platform Adverse Reactions and Precautions of Cetrorelix Acetate for Injection.
2.3 Improvement of Endometrial Receptivity and Enhancement of Embryo Implantation Rate
Embryo implantation is critical to successful assisted reproduction, and endometrial receptivity – the ability of the endometrium to support implantation – is the determining factor. Endometrial development must be precisely synchronized with embryonic development under the balanced regulation of estrogen and progesterone.
During COS, high-dose exogenous gonadotropins frequently cause excessive estrogen levels and premature progesterone elevation, disrupting synchrony between endometrial and embryonic development, reducing endometrial receptivity, and lowering implantation and clinical pregnancy rates.
It improves endometrial receptivity by fine-tuning hormonal levels, creating a more favorable environment for implantation. By inhibiting LH secretion, it reduces premature progesterone synthesis and release, delaying endometrial luteinization and aligning endometrial morphology, structure, and molecular markers (e.g., integrins, homeobox genes, leukemia inhibitory factor) with embryonic development. A stable hormonal milieu also reduces abnormal endometrial hyperplasia and vascular dysregulation, promoting normal glandular and stromal development and achieving an optimal implantation thickness of 8–12 mm.
Multicenter clinical studies show that cetrorelix-based antagonist protocols significantly improve endometrial receptivity compared with traditional GnRH agonist regimens, increasing implantation rates by 10%–15% and achieving clinical pregnancy rates above 55%. Benefits are particularly notable in patients with repeated implantation failure or poor endometrial receptivity. Cetrorelix acetate injection has no direct endometrial toxicity, and endometrial function recovers rapidly after cessation, supporting normal progression of subsequent pregnancies and confirming its safety and efficacy in ART.
Information source: Journal of Reproductive Medicine, 2025, clinical study data; Merck Healthcare Professionals Clinical Summary of Cetrorelix; Fast Doctor Clinical Value Analysis of Cetrorelix Acetate.
Applications in Other Potential Research Areas
Male Androgen-Related Diseases:The product can inhibit pituitary LH secretion in males and reduce testosterone levels, making it suitable for research on diseases associated with male androgen excess, such as benign prostatic hyperplasia (BPH) and gynecomastia. Clinical studies have demonstrated that daily subcutaneous injection of 0.25 mg leads to a significant decrease in male testosterone levels and alleviates symptoms in BPH patients including dysuria and frequent urination, providing a new direction for the pharmacotherapy of BPH.
Early Pregnancy Support in Assisted Reproduction:Several studies have explored the application value of this product in early pregnancy during assisted reproductive technology.

By suppressing fluctuations in LH levels, it stabilizes luteal function in early pregnancy and reduces the risk of early miscarriage. However, this field remains in the clinical research stage and has not been widely adopted in clinical practice; large-sample, multi-center Treatment of Rare Endocrine Diseases:For rare disorders characterized by abnormal hormone secretion (e.g., idiopathic precocious puberty and diseases related to excessive activation of GnRH receptors), elagolix acetate can block the GnRH signaling pathway, inhibit abnormal hormone secretion, and alleviate disease symptoms. Although relevant research is currently limited, its unique mechanism of action offers potential strategies for the treatment of rare endocrine diseases.
Information source: ClinicalTrials.eu. Cetrorelix [Online]; 2026,THE BioTek. Cetrorelix [Online]. 2025.
Adverse Reactions
Local reactions:
Erythema, pruritus, and swelling at the injection site (incidence approximately 9.4%).
Ovarian hyperstimulation syndrome (OHSS):
Mild to moderate (WHO Grade I or II) cases are common; severe cases are occasional.
Hypersensitivity reactions:
Including pseudoallergic/anaphylactoid reactions, rare but clinically important.
Information source:
Drug Instructions for Elagolix / Ganirelix Acetate; Public Clinical Safety Data from the European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA)
FAQ
What is cetrorelix acetate used for?
+
-
Cetrorelix is used to prevent premature ovulation as part of controlled ovarian stimulation treatment.
Is cetrorelix acetate the same as Cetrotide?
+
-
Cetrotide® (cetrorelix acetate for injection) is indicated for the inhibition of premature LH surges in women undergoing controlled ovarian stimulation. Cetrotide® (cetrorelix acetate for injection) should be prescribed by physicians who are experienced in fertility treatment.
Hot Tags: cetrorelix acetate injection, China cetrorelix acetate injection manufacturers, suppliers

























