Cetrorelix acetate 0.25 mg is a third-generation GnRH antagonist. This dosage is the clinical standard single daily dose, administered by subcutaneous injection once daily, with flexible dosing in the morning or evening. It facilitates patient self-administration and improves treatment compliance. With rapid onset, stable and controllable effects, it significantly reduces the risk of ovarian hyperstimulation syndrome (OHSS) and shortens the ovarian stimulation cycle. Local injection reactions are mild and transient, and it is well tolerated.
Our Products Form
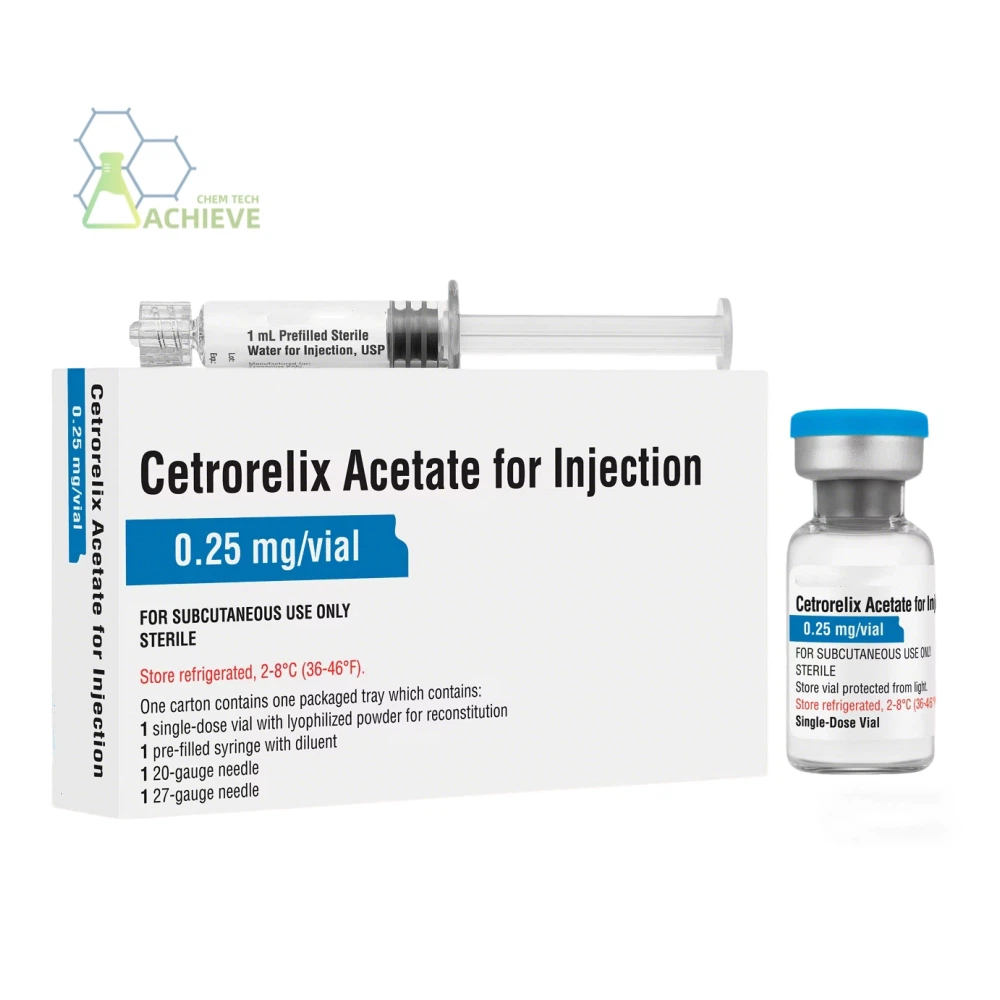


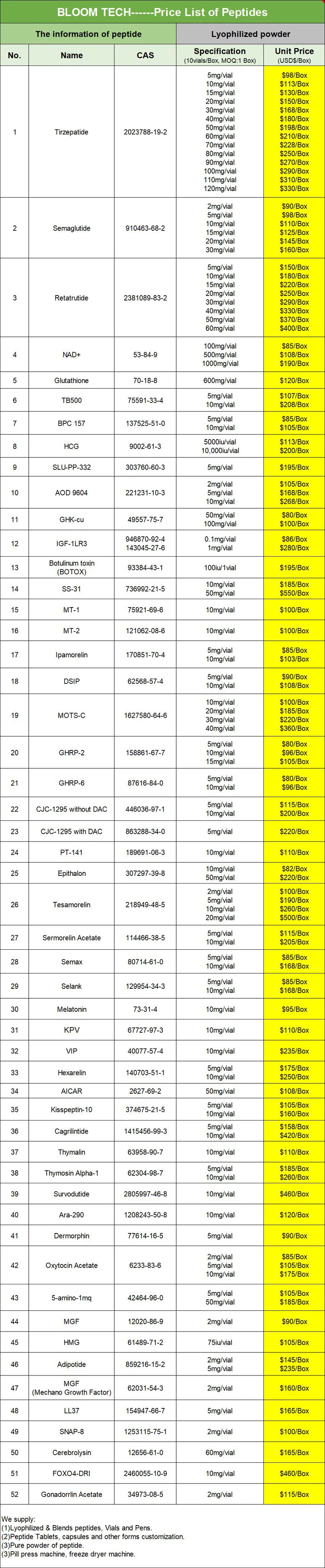


Cetrorelix acetate COA


Applications in Oncology
(I) Ovarian Cancer: Adjuvant Endocrine Therapy for Sex Hormone-Dependent Tumors
Ovarian cancer is a highly estrogen-dependent malignant tumor. GnRH antagonists can suppress the hypothalamic-pituitary-ovarian axis, reduce circulating estrogen levels, and block proliferative signaling in tumor cells.Cetrorelix acetate 0.25 mg administered subcutaneously once daily rapidly inhibits LH secretion, markedly reducing ovarian estrogen synthesis. Deprived of hormonal stimulation, tumor cells undergo growth arrest and apoptosis.
Clinically, it is mainly used as second-line endocrine therapy for advanced epithelial ovarian cancer, or in combination with chemotherapy and targeted agents for maintenance treatment in patients with platinum-resistant recurrent disease. It can reduce the tumor marker CA125 level and delay disease progression, especially suitable for elderly or frail patients who cannot tolerate intensive chemotherapy.Compared with GnRH agonists, the 0.25 mg formulation has no initial flare effect, takes effect within 4–12 hours after administration, avoids the risk of transient tumor proliferation, and offers better safety.

Information source:Chinese Expert Consensus on Endocrine Therapy for Ovarian Cancer (2024 Edition);Clinical trial data for expanded indications of the originator drug from EMD Serono.
(II) Prostate Cancer: An Important Option for Androgen Deprivation Therapy
Prostate cancer growth relies on testosterone, and androgen deprivation is the core treatment for locally advanced and metastatic prostate cancer.It administered subcutaneously once daily inhibits pituitary LH secretion, blocks testicular testosterone synthesis, and rapidly reduces serum testosterone to castrate levels (< 50 ng/dL).
Its mechanism is direct and reversible, without the drawback of initial testosterone elevation seen with agonists. Rapid castration is achieved within 3–5 days of treatment, significantly lowering prostate-specific antigen (PSA) levels, shrinking tumor volume, and relieving symptoms such as bone pain and dysuria.The daily 0.25 mg dose maintains stable hormonal suppression, avoiding fluctuations caused by high-dose intermittent administration. It is indicated for primary castration therapy in advanced prostate cancer, second-line endocrine therapy for castration-resistant prostate cancer, and preoperative tumor shrinkage to reduce surgical risk.
Information source:Chinese Guidelines for Diagnosis and Treatment of Urological and Andrological Diseases (2024 Edition);Controlled study of GnRH antagonists for prostate cancer indexed in PubMed (2023).
(III) Uterine Fibroids: Preoperative Preparation and Symptom Control
Uterine fibroids are estrogen-dependent benign tumors, in which a high-estrogen environment promotes fibroid cell proliferation and angiogenesis.Cetrorelix acetate 0.25 mg administered subcutaneously once daily, or a single 3 mg dose every 28 days combined with 0.25 mg maintenance therapy, rapidly suppresses estrogen secretion, placing fibroids in a low-hormone state and reducing volume by 30%–50%.
Clinically, it is mainly used for preoperative preparation, especially in patients with large fibroids (> 5 cm in diameter) accompanied by menorrhagia, anemia, or compressive symptoms. After 3–6 months of treatment, intraoperative blood loss is reduced and surgical difficulty is lowered; after fibroid shrinkage, minimally invasive surgery can be selected, shortening hospital stay.For perimenopausal patients, the 0.25 mg formulation can be used for long-term low-dose maintenance to relieve symptoms such as prolonged menstruation, heavy menstrual bleeding, and abdominal pain, delay fibroid progression, and avoid surgical trauma.
Information source:Chinese Expert Consensus on Diagnosis and Treatment of Uterine Fibroids (2025 Edition);Clinical research data from the Obstetrics and Gynecology Branch of the Chinese Medical Association.
(IV) Endometriosis: Lesion Atrophy and Pain Relief
The development and progression of endometriosis are closely related to estrogen, as ectopic endometrial tissue depends on estrogen for proliferation and invasion.The product administered subcutaneously once daily rapidly lowers estrogen levels, causing atrophy and necrosis of ectopic endometrial lesions, and significantly relieving core symptoms including dysmenorrhea, chronic pelvic pain, and dyspareunia.
Compared with traditional GnRH agonists, the 0.25 mg daily dose acts faster (estrogen inhibition within 24 hours of administration), has no flare effect, and causes milder hypoestrogenic symptoms (hot flashes, night sweats). It is suitable for short-term preoperative treatment (3 months) or long-term intermittent therapy.For patients with recurrent endometriosis, those intolerant to surgery, or for postoperative recurrence prevention, the 0.25 mg formulation can serve as a long-term management strategy, combined with add-back therapy (low-dose estrogen-progestin) to reduce side effects such as bone loss.
Information source:Guidelines for Diagnosis and Treatment of Endometriosis (2024);Summary of Product Characteristics (SPC) from the European Medicines Agency (EMA).
Applications in Benign Prostatic Hyperplasia (BPH)
Benign prostatic hyperplasia is a common disease in middle-aged and elderly men. Its progression is closely associated with dihydrotestosterone (DHT), which is converted from testosterone by 5α-reductase. Chronic stimulation by DHT leads to prostatic cell hyperplasia, resulting in urethral compression and voiding dysfunction.
Cetrorelix acetate 0.25 mg administered subcutaneously once daily inhibits pituitary LH secretion, reduces testicular testosterone production, lowers DHT synthesis at the source, and causes atrophy and volume reduction of hyperplastic prostatic tissue.
Clinically, the 0.25 mg formulation is mainly used as adjuvant therapy for patients with moderate to severe BPH, especially those with elevated PSA and suspected prostate cancer risk, or those intolerant to 5α-reductase inhibitors (e.g., finasteride).
After 3–6 months of treatment, the International Prostate Symptom Score (IPSS) is significantly improved, relieving symptoms such as urinary frequency, urgency, dysuria, and nocturia, increasing maximum urinary flow rate, and reducing the risk of acute urinary retention.When combined with α-blockers, 0.25 mg provides long-term stable hormonal suppression, preventing symptom rebound and supporting long-term conservative management of BPH.The daily 0.25 mg dose delivers steady hormonal control with no significant side effects such as sexual dysfunction or breast tenderness, resulting in better patient compliance than high-dose GnRH antagonists.

Information source:Guidelines for Diagnosis and Treatment of Benign Prostatic Hyperplasia (2024 Edition);Indication records from Hunan Pharmaceutical Affairs Service Network.
Manufacturing Information
I. Core Synthetic Process: Solid-Phase Peptide Synthesis (Fmoc Strategy)
Ovurelix is manufactured via Fmoc solid-phase peptide synthesis, the mainstream industrial production route. Using Rink Amide-AM resin as the carrier, synthesis starts from the C-terminal D-alanine, with sequential coupling of Fmoc-protected amino acids (including non-natural amino acids such as D-2-Nal, D-4-Cl-Phe, D-3-Pal). The Fmoc protecting group is removed with piperidine at each step, using HATU/HOBt as coupling agents, with strict control of racemization (arginine and serine isomer impurities <0.5%). The N-terminus is directly coupled with Ac-D-2-Nal-OH or acetylated to avoid side-chain side reactions. Cleavage is performed with 95% aqueous trifluoroacetic acid (TFA) at room temperature for 60–90 minutes; crude peptide purity ≥60% is required for purification.
Information source: Technical Specification for Polypeptide API Production: Cetrorelix Acetate T/SZLSBA 001-2019
II. Purification and Salt Conversion: Key to High-Purity Control
The crude peptide is dissolved in glacial acetic acid, filtered, and purified by preparative reversed-phase high-performance liquid chromatography (RP-HPLC) with gradient elution to separate impurities and isomers. A C18 column and acetic acid/acetonitrile system are used to precisely collect the main peak and remove close-eluting impurities such as D-arginine and D-serine. After purification, reverse osmosis concentration and salt conversion are performed to convert the trifluoroacetate salt to acetate, eliminating TFA residues. A second refining step and sterile filtration follow, ensuring final product purity ≥99.5% and single impurity <0.1%.
Information source: Patent CN112250735-B, PubMed 2025 synthesis studies
III. Formulation Manufacturing: Precise Preparation of 0.25 mg Lyophilized Powder for Injection
The active pharmaceutical ingredient (API) is mixed with mannitol, acetic acid and other excipients in proportion, dissolved in water for injection to form the drug solution. After 0.22 μm sterile filtration, the solution is accurately filled into vials at 0.25 mg per vial. Vacuum freeze-drying is applied: pre-freezing at -45°C for 8 hours, followed by primary sublimation and secondary desorption drying, with residual moisture controlled to <3.0%. The entire process is conducted in Grade C + Grade A clean areas under nitrogen protection to prevent oxidation. Finished products undergo testing for content, purity, sterility and endotoxins, complying with registration standard JX20130315.
Information source: National Medical Products Administration drug standards, Livzon Pharmaceutical manufacturing process
IV. Quality Control and Compliance: Strict Full-Process Standards
Production complies with multiple standards including the Chinese Pharmacopoeia, European Pharmacopoeia (EP) and United States Pharmacopeia (USP). Key quality control items:① Amino acid composition: within ±2% of theoretical value;② Assay: 95.0%–105.0%;③ Related substances: single impurity <0.1%, total impurities <1.0%.
Ultra-high performance liquid chromatography (UPLC) is used to detect six characteristic impurities, with a detection limit as low as 0.04 ng. Manufacturing requires GMP certification, with complete batch traceability and full-chain validation from raw materials to finished products.
Information source: Chinese Journal of Modern Applied Pharmacy, 2022; EP 11.0
Contraindications
Hypersensitivity to Ovurelix, GnRH analogues, exogenous peptide hormones or any excipients.
Patients with moderate to severe hepatic or renal impairment (contraindicated in severe renal impairment).
Information source:Originator prescribing information, EMD Serono (US); Summary of Product Characteristics (SPC), European Medicines Agency (EMA)
Hot Tags: cetrorelix acetate 0.25 mg, China cetrorelix acetate 0.25 mg manufacturers, suppliers