SLU-PP-332 tablet is an oral small molecule targeted therapeutic drug in the preclinical or early clinical research stage. The core active ingredient SLU-PP-332 is designed to specifically inhibit certain proteins or signaling pathways that play crucial roles in the proliferation and survival of cancer cells, possibly involving epigenetic regulation or transcription processes, with the aim of interfering with tumor growth. This tablet is produced using standardized solid formulation technology, aiming to provide a stable dose and good oral bioavailability, making it convenient for patients to take. Currently, the detailed pharmacological mechanism, indication range, optimal dosage, and safety data of this drug have not been fully disclosed and are still under strict laboratory and animal model validation. The main evaluations include its anti-tumor efficacy, pharmacokinetic characteristics, and preliminary toxicity. The research goal is to achieve effective control of specific types of cancer through precise targeting and lay the foundation for subsequent human clinical trials.
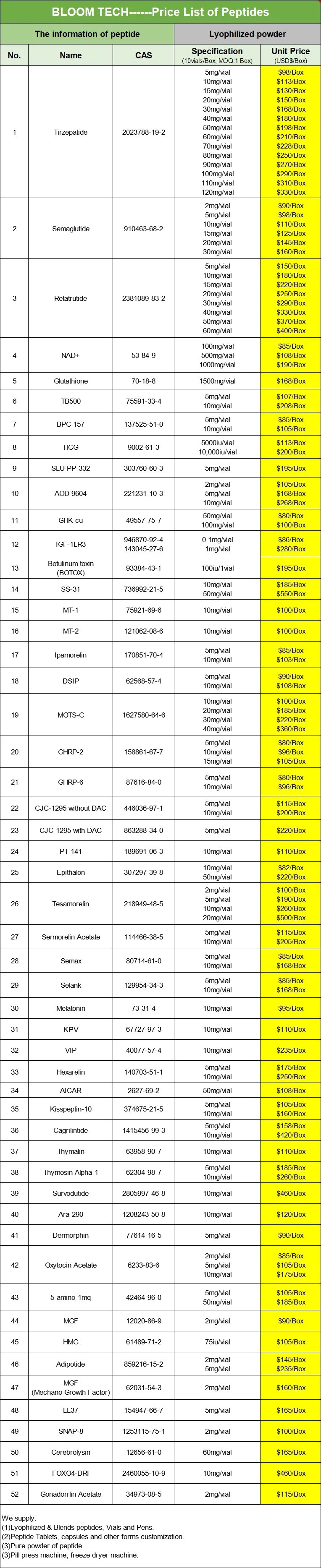
Our products form






SLU-PP-332 COA


Stable formula enhances treatment compliance
In the field of metabolic disease treatment, patient compliance has always been a key factor influencing the therapeutic effect. Traditional drugs often have obvious side effects, complex dosing regimens, or require strict dietary and exercise plans, making it difficult for patients to adhere to the treatment for a long time. However, the oral solid formulation of SLU-PP-332 tablets, with its stable formula and unique mechanism of action, provides an innovative solution to enhance treatment compliance, redefining the convenience and sustainability of metabolic health management.




Stable formula: Ensuring efficacy and reducing dosage fluctuations
The oral solid formulation of SLU-PP-332 uses high-purity raw materials (purity > 98%) and advanced formulation technology to ensure the uniform distribution of active ingredients in each tablet. Its stable formula can be stored for a long time at room temperature, avoiding drug degradation due to environmental factors, thereby ensuring the continuity of efficacy. This stability not only reduces the inconvenience for patients who need to frequently switch medications due to drug expiration, but also lowers the risk of side effects caused by inaccurate dosing, providing a reliable guarantee for long-term treatment.
Non-dietary Dependency: Breaking the Limitations of Traditional Weight Loss Drugs
Traditional weight loss drugs usually work by suppressing appetite or blocking fat absorption. However, these drugs often come with side effects such as malnutrition and gastrointestinal discomfort, and patients need to strictly control their diet, resulting in low compliance. SLU-PP-332, on the other hand, activates the ERR receptor to simulate the effect of exercise, promoting fat breakdown for energy supply, without the need to suppress appetite or change the diet structure. Experimental data shows that obese mice lost 12% of their weight after 28 days of medication, and their food intake was not affected. This "unburdened weight loss" mechanism enables patients to avoid struggling between hunger and health, significantly improving the acceptance of treatment.
Long-term Mechanism: Simplified Medication Regimen, Enhanced Convenience
The oral solid formulation of SLU-PP-332 is designed for a once-daily dosing frequency. Its long-lasting mechanism of action can maintain metabolic regulatory effects for 24 hours. This simplified regimen avoids the complexity of multiple doses and is particularly suitable for individuals with busy schedules or memory decline. For instance, in normal-weight mice, after administration of SLU-PP-332, the running time increased by 70% and the distance increased by 45%, without the need to increase the exercise frequency. This characteristic indicates that the drug can achieve a "passive exercise" effect by continuously activating metabolic pathways, reducing the burden on patients to actively perform high-intensity training.
Safety Tolerance: Reducing the Risk of Side Effects and Enhancing Treatment Confidence
Short-term preclinical safety studies have shown that SLU-PP-332 is well-tolerated at effective doses in rodents, with no reports of serious adverse events. Its characteristic of selectively activating the ERR receptor avoids the side effects (such as hormonal disorders or organ toxicity) caused by off-target effects of early ERR agonists. This safety advantage makes patients more willing to adhere to treatment for a long term rather than stopping treatment halfway due to fear of side effects. For example, in the heart failure model, SLU-PP-332 not only improved cardiac function but also increased survival rate, further verifying its potential for clinical application.
Patient Support System: Comprehensive Enhancement of Compliance
Apart from the inherent characteristics of the drug itself, the treatment plan of SLU-PP-332 also incorporates a patient support system. Through tools such as smart pill boxes and mobile apps for reminders, patients can easily manage their medication times and avoid missing doses or taking the wrong ones. At the same time, doctors and pharmacists can monitor the patient's treatment efficacy through remote follow-ups, adjust the plan in a timely manner, and answer questions. This "drug + service" integrated model strengthens the patient's treatment motivation at the psychological and behavioral levels, making metabolic health management a sustainable lifestyle.

SLU-PP-332 tablets,as a new type of oral solid formulation, has its core advantage in mimicking the effects of exercise by activating estrogen-related receptors (ERRs), providing a non-dietary and non-exercise-dependent solution for the treatment of metabolic diseases. The stability and safety design of this product runs through the entire research and development process, from the optimization of chemical structure to the improvement of formulation technology, and to the preclinical safety assessment, all of which reflect a deep consideration for long-term efficacy and patient tolerance.
Stability Design: Ensuring throughout the entire chain from molecules to formulations
The stability design of SLU-PP-332 focuses on three key dimensions:
Chemical structure stability
As a broad ER agonist, the E-oxime configuration of SLU-PP-332 is the core structure for its binding to the receptor. Through optimization of the synthesis process, this configuration is ensured to exist stably at normal temperature and pressure, avoiding the loss of activity due to configuration isomerization. Preclinical studies have shown that its purity can be stably maintained above 98%, providing a molecular basis for long-term efficacy.
Formulation process stability
The oral solid formulation adopts rapid-release technology. By adding solvents for quick dissolution and optimizing the particle structure, the drug rapidly disintegrates in the gastrointestinal tract, ensuring the efficient release of the active ingredient. At the same time, the solid form effectively isolates the effects of light, heat, and humidity, allowing the drug to be stably stored for 3 years at normal temperature, avoiding the impact of degradation on safety. The packaging design for dispensing further reduces the loss of activity due to repeated freezing and thawing, ensuring that each tablet can achieve the expected therapeutic effect.
Storage and transportation stability
For the special nature of oral formulations, the storage conditions of SLU-PP-332 are set as a cool, dry, and well-ventilated environment, avoiding direct sunlight. During transportation, blue ice/normal temperature packaging is used to ensure that the drug remains active and stable before reaching the patient. This stability design not only extends the drug's shelf life but also reduces the risk of efficacy fluctuations due to improper storage.
Safety assessment: Comprehensive verification from mechanism to clinical
The safety assessment system of SLU-PP-332 covers three levels: mechanism analysis, animal experiments, and preclinical studies:

Mechanism safety
SLU-PP-332 selectively activates the ERRα/β/γ receptors, simulating the metabolic adaptive changes during physical activity, such as increasing fatty acid oxidation, reducing carbohydrate utilization, and enhancing glucose uptake. Its action pathway is independent of the appetite suppression mechanism of traditional weight loss drugs, avoiding the risk of malnutrition. At the same time, the drug design fully considers off-target effects, reducing the impact on non-target organs such as the reproductive system and cardiovascular system through structural optimization.
Animal experiment safety
In the obesity mouse model, after a single course of treatment with SLU-PP-332 tablets for 28 days, the body weight decreased by 12%, and liver fat, cholesterol, and triglycerides were significantly reduced. No serious adverse reactions were reported. In normal mice, the running time increased by 70% and the distance increased by 45% after medication, while exercise endurance improved, and appetite and autonomous movement intention were not affected. In the heart failure mouse model, the drug improved cardiac function, reduced fibrosis, and increased survival rate, further verifying its tissue-specific safety.


Preclinical safety data
Short-term preclinical studies have shown that SLU-PP-332 has good tolerance at effective doses in rodents and no serious adverse events were observed. However, comprehensive toxicological studies, pharmacokinetics, off-target analysis, and long-term safety data still need to be determined. Currently, the drug is still in the preclinical research stage and needs to be further evaluated for its safety profile and potential side effects through strict human clinical trials.

Synergistic effect of stability and safety
The stability and safety design of SLU-PP-332 form a synergistic effect: The stable chemical structure and formulation process ensure the continuous release of active ingredients in the body, maintaining a stable blood drug concentration, thereby reducing the risk of side effects caused by drug fluctuations; Precise mechanism regulation and comprehensive safety assessment provide scientific basis for its long-term use, especially suitable for people with health limitations who cannot exercise. This synergistic effect makes SLU-PP-332 an important candidate drug in the field of metabolic disease treatment, potentially redefining the convenience and sustainability of health management.
FAQ
1. What disease does SLU-PP-332 tablets treat?
Currently, it is still in the preclinical research stage and the specific indications have not been determined. It is a targeted anti-cancer drug, mainly targeting certain cancers that rely on specific signaling pathways. However, which specific type of cancer it treats needs to be verified through subsequent clinical trials.
2. What is its main mechanism of action?
It is an oral small molecule targeted drug. Its core mechanism is to specifically inhibit key proteins (such as certain transcriptional regulators) that are closely related to the proliferation and survival of tumor cells, thereby blocking the growth signals of cancer cells and inducing their death.
3. What is SLU-PP-332?
It is an oral targeted anti-cancer tablet in the pre-clinical research stage, designed to function by specifically inhibiting key proteins related to tumor growth.
Hot Tags: slu-pp-332 tablets, China slu-pp-332 tablets manufacturers, suppliers, AOD 9604 Capsules, AOD 9604 Powder, Bioglutide NA 931 Capsules, bodybuilding peptide, Semaglutide Tablets 7mg, SLU PP 332 Powder












