Cetrorelix Tablet are oral formulations of cetrorelix, specifically designed for controlled ovulation in assisted reproductive technology. Distinguished from conventional injectable forms, this oral preparation improves patient compliance. Featuring precise dosing and convenient administration without injection, it is suitable for long-term outpatient use.
Our Products Form




Cetrorelix COA
 |
||
| Certificate of Analysis | ||
| Compound name | Cetrorelix | |
| Grade | Pharmaceutical grade | |
| CAS No. | 120287-85-6 | |
| Quantity | 51g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090056 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.31% |
| Loss on drying | ≤1.0% | 0.23% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.80% |
| Single impurity | <0.8% | 0.32% |
| Total microbial count | ≤750cfu/g | 337 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 516ppm |
| Storage | Store in a sealed, dark, and dry place below 2-8°C | |
|
|
||
|
|
||
| Chemical Formula | C70H92ClN17O14 |
| Exact Mass | 1429.67 |
| Molecular Weight | 1431.06 |
| m/z | 1429.67(100.0%), 1430.67(75.7%), 1431.67(32.0%), 1431.68(28.3%), 1432.67(24.2%), 1433.67(9.0%), 1430.67(6.3%), 1432.68(6.1%), 1431.67(4.5%), 1431.67(2.9%), 1434.68(2.2%), 1432.68(2.2%), 1432.66(1.9%), 1432.67(1.8%), 1433.67(1.5%), 1433.68(1.3%), 1430.68(1.1%) |
| Elemental Analysis | C,58.75; H,6.48; Cl,2.48; N,16.64; O,15.65 |

Cetrorelix tablet have been widely used in the multi‑stage and multi‑scenario treatment of prostate cancer owing to their advantages of rapid androgen deprivation, no hormone flare, and high safety. They cover palliative therapy for advanced disease, neoadjuvant/adjuvant therapy for locally advanced disease, and salvage therapy for biochemical recurrence after surgery or radiotherapy.
It is indicated for patients with hormone-sensitive advanced prostate cancer who are ineligible for surgical resection or radiotherapy, or who have developed bone, lymph node, or visceral metastases. It can be used as a single endocrine therapy or as a foundational agent in combination regimens. Its core value lies in rapidly controlling tumor progression, relieving clinical symptoms, and prolonging patient survival.
For patients with advanced prostate cancer, especially those complicated by acute conditions such as spinal cord compression, urinary tract obstruction, or severe bone pain, cetrorelixum demonstrates distinct superiority: it lacks an initial testosterone surge effect, thereby avoiding exacerbation of tumor symptoms caused by transient testosterone elevation, including aggravated bone pain, worsened spinal cord compression, and increased hematuria, while reducing the risk of severe complications. In clinical practice, after administration of cetrorelixum, significant symptomatic relief occurs within two weeks in advanced patients. The remission rate of bone pain exceeds 70%, and the improvement rate of dysuria, hematuria and other symptoms is above 65%. Meanwhile, it rapidly reduces prostate-specific antigen (PSA) levels, buying time for subsequent treatments.
Neoadjuvant therapy: For patients with locally advanced prostate cancer (clinical stage T3–T4), large tumor volume, extra-capsular extension, or high risk of lymph node metastasis, cetrorelixum administered for 4–8 weeks preoperatively can rapidly shrink tumor volume, downstage the disease, reduce the scope of tumor invasion, and improve the success rate of radical prostatectomy.
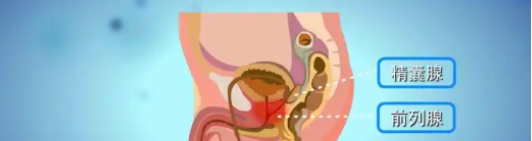
Adjuvant therapy: For patients with high-risk recurrence factors after radical prostatectomy or radical radiotherapy, such as positive surgical margins, capsular invasion, seminal vesicle involvement, or lymph node metastasis, long-term postoperative or post-radiotherapy use of cetrorelixum maintains castrate levels of testosterone, eliminates minimal residual disease, and reduces the risk of biochemical recurrence and distant metastasis.
Biochemical recurrence is a common clinical event after radical prostatectomy or radiotherapy for prostate cancer, defined as a persistent rise in PSA (>0.2 ng/mL) without evidence of radiological metastasis. Timely initiation of salvage endocrine therapy at this stage can effectively delay tumor progression and prevent progression to metastatic prostate cancer. As a first-line option for salvage endocrine therapy, cetrorelixum rapidly suppresses testosterone levels, controls PSA progression, and delays metastasis.

Compared with continuous long-term administration, cetrorelix tablet can be used in an intermittent regimen: treatment is suspended when PSA decreases to an extremely low level (<0.1 ng/mL) and resumed when PSA rises above 0.2 ng/mL. This regimen reduces adverse reactions associated with long-term use, preserves sexual function and quality of life, and maintains therapeutic efficacy. It is particularly suitable for younger patients with biochemical recurrence who have high quality-of-life requirements.
Sources of Information:AUA Guidelines, Hormonal Therapy for Prostate Cancer (2026); Nootroholic, Cetrorelixum Combination Therapy Clinical Review (2026); The Journal for ImmunoTherapy of Cancer, Synergistic Potential of Immunotherapy in mCRPC (2026).
1. Research and Development Background
The discovery of cetrorelixum stemmed from breakthroughs in basic research on gonadotropin-releasing hormone (GnRH). In 1971, Schally's team successfully isolated and characterized the decapeptide structure of native GnRH, laying the foundation for subsequent analog development. Early clinical practice relied on GnRH agonists, which exhibited an initial hormone flare-up effect and failed to meet the need for rapid LH surge suppression. Meanwhile, first- and second-generation GnRH antagonists were limited clinically by severe adverse effects such as edema and hypersensitivity due to histamine release. Against this background, third-generation GnRH antagonists with high activity and minimal side effects became the core focus of research.
2. Molecular Discovery and Structural Optimization (1988–1990)
In 1988, the team led by Nobel laureate Schally launched the development of novel GnRH antagonists. In 1990, Bokser et al. at Tulane University (USA) first synthesized cetrorelixum, a synthetically modified decapeptide containing five non-natural D-amino acids, which markedly enhanced receptor affinity and metabolic stability. Through multiple rounds of amino acid substitution, the research team completely eliminated histamine release-related side effects while retaining potent competitive antagonistic activity at pituitary GnRH receptors, enabling rapid and reversible suppression of LH and FSH secretion. Preclinical studies confirmed its safe and effective inhibition of gonadotropin secretion in various animal models, paving the way for clinical trials.
In 1994, cetrorelixum became the first GnRH antagonist to enter Phase III clinical trials, focusing on verifying its efficacy in preventing premature LH surges during assisted reproductive COS. It was approved by the European Medicines Agency (EMA) in April 1999 as the first third-generation GnRH antagonist for clinical use in Europe, and by the U.S. Food and Drug Administration (FDA) in August 2000 under the brand name Cetrotide. The originator product entered China in 2010 under the brand name Sizekai. Its development overcame the flare-up drawback of agonists and side effects of early antagonists, marking a milestone drug in assisted reproduction.

4. Developmental Significance and Academic Value
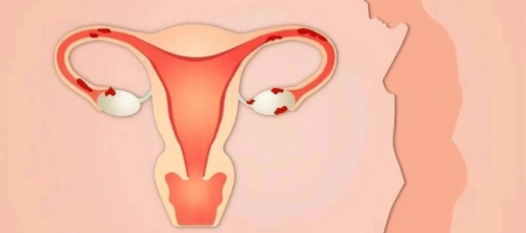
The discovery of cetrorelixum signaled the maturation of GnRH antagonist development from basic research to clinical application, establishing a third-generation antagonist design paradigm characterized by high receptor affinity, low histamine release, and rapid inhibition. Its developmental pathway served as a template for subsequent agents such as ganirelix and degarelix, driving therapeutic innovations in assisted reproduction, gynecological disorders, and oncofertility. As the first commercially successful potent GnRH antagonist, cetrorelixum's development history exemplifies the core value of translating basic medical science into clinical therapy.
Sources of Information:Kutscher H, et al. The LHRH antagonist Cetrorelixum. Hum Reprod Update. 2000;6(4):322–331; Engel J, Reissmann T. Cetrorelixum, a potent LHRH-antagonist: Chemistry, pharmacology and clinical data; Arzneim Forsch. 1999;49(8):697–705.Chinese Society of Reproductive Medicine; Clinical Practice Guidelines for Assisted Reproductive Technology (2022); Chinese Journal of Reproduction and Contraception, 2022; Lambertini M, et al. Fertility preservation in young women with early breast cancer. J Clin Oncol. 2016;34(25):3035–3044.
1. Solubility and Acid-Base Properties
Cetrorelix Tablet has low water solubility (approximately 2.0 mg/mL), is freely soluble in DMSO (solubility increases with heating), and slightly soluble in methanol. Formulation optimization with excipients improves in vivo dissolution and absorption stability of the tablets. With a pKa ≈ 9.82, it is a weakly basic polypeptide with optimal stability in the pH range of 5–8. It exists as a zwitterion under neutral conditions, with a plasma protein binding rate of approximately 86%.
2. Stability and Degradation Characteristics
It are sensitive to light, heat, strong acids, and strong bases, requiring refrigerated storage at 2–8°C in a light-protected, sealed container. Both the drug substance and tablets have a shelf life of up to 3 years. Aqueous solutions are stable for 21 days at pH 7.0 and room temperature; rapid hydrolysis occurs under high temperature (100°C) or extreme pH conditions, with approximately 15% degradation within 30 minutes. The D-amino acids in the structure significantly enhance resistance to enzymatic hydrolysis, with tolerance to chymotrypsin, pronase, and other enzymes exceeding 50 hours, far superior to native GnRH and agonists.
3. Chemical Characteristics of the Tablet Formulation
It use the acetate salt as the active ingredient, combined with excipients including microcrystalline cellulose, lactose, and magnesium stearate to ensure uniform content, rapid disintegration, and stable oral absorption. The formulation lacks histamine-releasing side chains, eliminating the hypersensitivity risk of early antagonists. Rapid absorption after oral administration results in bioavailability comparable to injectable formulations, with reversible action and no receptor desensitization, meeting clinical needs for rapid LH surge suppression and flexible cycle regulation.
Sources of Information:Engel J, Reissmann T. Cetrorelixum, a potent LHRH-antagonist: Chemistry, pharmacology and clinical data. Arzneim Forsch. 1999;49(8):697–705.National Medical Products Administration. Drug Registration Standard for Cetrorelixum. 2023.
Medical monitoring for 30 minutes is required after the first dose to detect hypersensitivity reactions.
Contact a physician immediately if a dose is missed; do not adjust the dosage independently.
Contraindicated in pregnant women (high-dose animal studies show embryo resorption and post-implantation loss).

Sources of Information:European Medicines Agency (EMA). Cetrotide (cetrorelixum) Summary of Product Characteristics (SmPC). 2024.
FAQ
When do you take it during IVF?
+
-
Cetrotide 0.25 mg may be administered subcutaneously once daily during the early- to mid-follicular phase. Cetrotide 0.25 mg is administered on either stimulation day 5 (morning or evening) or day 6 (morning) and continued daily until the day of hCG administration.
What is the product success rate?
+
-
The mean duration of cetrorelix treatment was 5.7 days. Success rate, defined as absence of LH surge, was 95.2% for CET group and 98% of patients on buserelin.
Hot Tags: cetrorelix tablet, China cetrorelix tablet manufacturers, suppliers