As a highly representative synthetic peptide member of the gonadotropin-releasing hormone (GnRH) agonist family, the action trajectory of triptorelin acetate 3.75 mg in vivo is not a single inhibitory effect, but presents a unique three-phase evolution process with clear temporal sequence, which is also the core logic of its pharmacological effects. Specifically, after the drug enters the body, it briefly activates the pituitary GnRH receptor as an agonist in the initial stage, triggering a transient release peak of gonadotropins (mainly luteinizing hormone LH and follicle stimulating hormone FSH), which is known as the flare up effect; As medication continues, pituitary cells will adaptively regulate the long-term activation of GnRH receptors, gradually entering a state of pituitary desensitization.
Products Description
 |
|



Triptorelin Acetate COA
 |
||
| Certificate of Analysis | ||
| Compound name | Triptorelin Acetate | |
| Grade | Pharmaceutical grade | |
| CAS No. | 140194-24-7 | |
| Quantity | 38g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090068 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
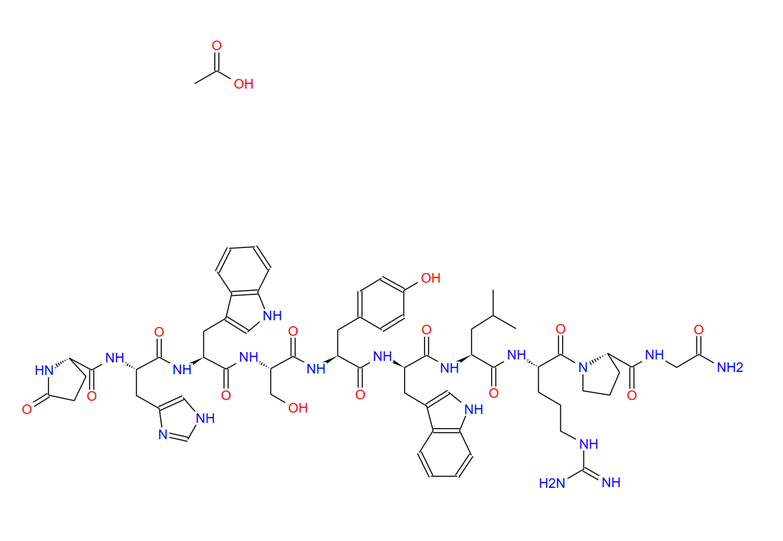
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.54% |
| Loss on drying | ≤1.0% | 0.42% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.98% |
| Single impurity | <0.8% | 0.52% |
| Total microbial count | ≤750cfu/g | 95 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 500ppm |
| Storage |
Store in a sealed, dark, and dry place below -20°C |
|
|
|
||
|
|
||
| Chemical Formula | C64H82N18O13 |
| Exact Mass | 1310.63 |
| Molecular Weight | 1311.47 |
| m/z | 1310.63 (100.0%), 1311.63 (69.2%), 1312.64 (23.6%), 1311.63 (6.6%), 1313.64 (4.5%), 1312.63 (4.3%), 1312.64 (2.7%), 1313.64 (1.8%), 1313.63 (1.6%) |
| Elemental Analysis | C, 58.61; H, 6.30; N, 19.22; O, 15.86 |

The ultimate outcome of pituitary desensitization is the significant inhibition of gonadotropin secretion, which leads to a sustained decrease in testosterone (male) or estrogen (female) levels in the body, ultimately reaching castration or postmenopausal levels. This complete sequence of action chain is not an isolated three stages, but an interconnected and progressive organic whole, which together constitute the core pharmacological basis of the clinical application of it and its key mechanism of action for the treatment of related diseases. In the following article, we will combine the characteristics of molecules, cells, and the whole body to conduct a multidimensional detailed disassembly and analysis of these three key pharmacological links.
Initial transient stimulation of gonadotropin release (flare up effect)
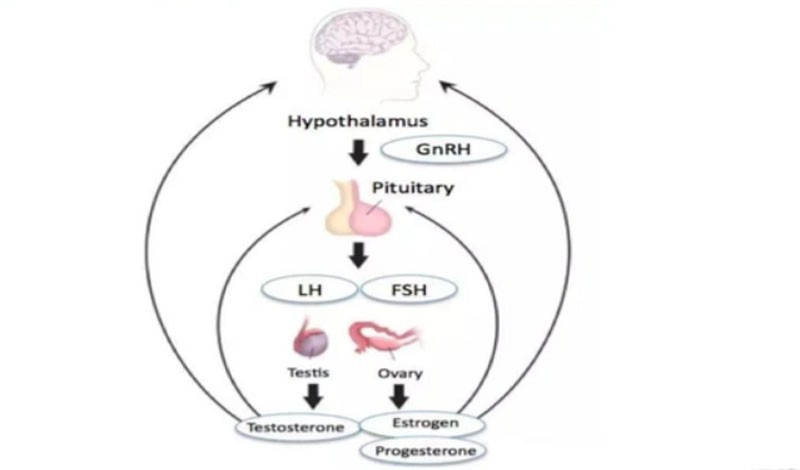
The initial effect of triptorelin acetate 3.75 mg is not direct inhibition, but rather activation of pituitary GnRH receptors as an agonist, triggering a brief surge in gonadotropins, known as the flare up effect. As a synthetic GnRH analogue, its 6th D-tryptophan replaces the L-glycine of natural GnRH. This structural modification not only significantly enhances its affinity with the receptor, making its affinity 50-100 times that of natural GnRH, but also endows it with stronger enzymatic resistance, extending the half-life from minutes to hours of natural hormones, providing a solid foundation for subsequent sustained action; Unlike the pulsatile regulation of natural GnRH, it breaks the physiological rhythm in a non pulsatile sustained binding mode, triggering receptor overactivation and opening the molecular basis of the flare up effect.From the perspective of hormone release kinetics, the peak blood concentration of the drug can be reached about 40 minutes after subcutaneous injection.
Within the following 24-72 hours, the level of luteinizing hormone (LH) can rise to 5-8 times the baseline value, and follicle stimulating hormone (FSH) will also increase synchronously but slightly less, reflected in the level of sex hormones. Male testosterone can briefly increase by 50-100%, and female estrogen levels will also show a corresponding upward trend. This effect usually lasts for 1-2 weeks before entering a rapid decline period, completing the transition from stimulation to transition. At the cellular signaling mechanism level,this drug activates Gq/11 protein coupled receptors, initiates the phospholipase C pathway, promotes the release of intracellular calcium stores, and triggers the exocytosis of gonadotropin storage granules, achieving rapid release of LH and FSH.
At the same time, it briefly upregulates the transcription of gonadotropin genes, further enhancing hormone synthesis rate and improving the regulation process of flare up effect at the cellular level. Although this transitional stage lays the foundation for subsequent pharmacological effects, there are also certain clinical potential risks. In the treatment of prostate cancer, temporarily elevated testosterone may worsen bone pain or urinary tract obstruction. Therefore, it is necessary to be alert to this risk and combine anti androgen drugs for prevention; Female patients may experience brief bleeding or worsening symptoms during this stage. In clinical practice, patients should be informed in advance and monitored to ensure medication safety.
Data source:
An Update on Triptorelin: Current Thinking on Androgen Deprivation Therapy for Prostate Cancer. PMID: 27375389
Effects of long-term treatment with GnRH agonists on pituitary function recovery. Fertil Steril, 2017
Pituitary desensitization and significant decrease in LH and FSH after continuous medication
When drugs are continuously present at therapeutic doses, the long-term activation of GnRH receptors by pituitary cells undergoes adaptive changes, known as desensitization, which ultimately leads to significant inhibition of gonadotropin secretion and becomes the core transitional link for drugs to exert therapeutic effects. At the receptor level, GnRH receptors that are continuously occupied by drugs undergo conformational changes, leading to receptor internalization and lysosomal degradation, resulting in a 70-80% decrease in receptor density on the cell surface.At the same time, receptor mRNA expression is downregulated, and the synthesis of new receptors is blocked, resulting in a state of "receptor depletion"; More importantly, the G protein coupled signaling pathway undergoes uncoupling, and even if the receptor and ligand can still bind.
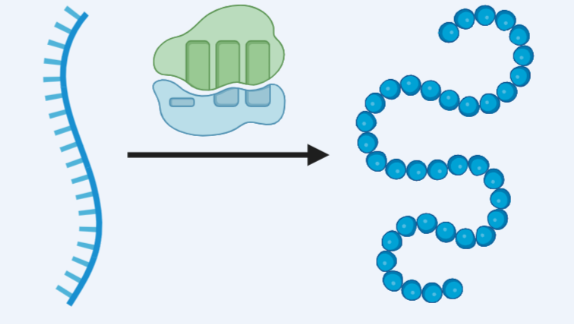
It cannot effectively transduce signals, blocking the signal transmission of gonadotropin secretion at the receptor level.The changes at the receptor level further trigger the remodeling of pituitary cell function, and gonadotropin cells enter a "refractory period", with a sharp decrease in responsiveness to subsequent stimuli. The coupling mechanism between hormone synthesis and secretion is disrupted, resulting in a decrease in LH and FSH release levels below 10-20% of baseline. At the same time, MAPK phosphatase induction increases, inhibiting signal cascade amplification, forming a sustained inhibitory state, ensuring stable decline in gonadotropin levels.From the perspective of time effect relationship, the desensitization process usually begins in the second week of medication.And a significant decrease in LH/FSH levels can be observed within 10-15 days of medication.
A stable inhibitory state can be achieved around 4 weeks of medication, at which point LH levels often drop to undetectable levels.There are differences in the duration of desensitization between different dosage forms of drugs. Long acting formulations (such as 3.75mg microspheres) can maintain a stable desensitization state for up to 30 days, while the 22.5mg dosage form can last for 6 months, meeting the needs of different clinical treatment scenarios. In addition, the sustained inhibitory effect will also break the negative feedback regulation of the hypothalamic pituitary gonadal axis, forming a "drug-induced hypogonadotropin state". At the same time, the sensitivity of the pituitary gland to feedback inhibition of sex hormones is reduced, further consolidating the inhibitory effect of gonadotropins and providing guarantees for the subsequent deep decline of sex hormone levels.
Data source:
Gonadotropin-releasing hormone (GnRH) agonists. UpToDate, 2023
Pituitary and extrapituitary actions of gonadotrophin-releasing hormone and its analogues. Hum Reprod, 1999
Gonadotrophin-releasing hormone and GnRH agonists: mechanisms of action. PubMed, 2003

The three-phase pharmacological effects of triptorelin acetate 3.75 mg are not isolated stages, but a complete pharmacological system that is closely connected, progressive, and interlocking. This unique mode of action has become its core clinical advantage compared to other types of hormone regulating drugs, demonstrating its unique value in the treatment of hormone dependent diseases.
Among them, the flare up effect, as the initial transitional stage of the entire pharmacological process, although only manifested as a brief peak of gonadotropin release, clearly reveals its essential properties as a GnRH agonist - it does not directly exert inhibitory effects, but regulates through the pathway of activating receptors first and then inducing subsequent adaptive changes. This initial stimulation process is not only a sign of drug action initiation, but also lays the necessary physiological foundation for subsequent pituitary desensitization.Without this brief excitatory effect, the subsequent desensitization process cannot be smoothly initiated. Pituitary desensitization, as a key intermediate link between initial stimulation and final therapeutic effect.
Plays a core role in inducing a series of adaptive changes in pituitary cells through continuous medication, including receptor depletion, signal pathway uncoupling, etc.,thereby completely blocking abnormal secretion of gonadotropins. The stability and persistence of regulation in this link directly determine the overall therapeutic effect of the drug and is the core of achieving clinical treatment goals. The reduction of sex hormone levels to castration (male) or postmenopausal (female) levels is the ultimate endpoint of the entire pharmacological action chain and the direct source of clinical benefits. By precisely controlling the level of sex hormones in the body at a low pathological level, it can effectively block the growth and progression of hormone dependent lesions, alleviate disease-related symptoms, and provide strong pharmacological support for the clinical treatment of hormone dependent diseases.

References
Triptorelin Acetate Microspheres for Prostate Cancer: Pharmacokinetics and Efficacy. Clin Pharmacokinet, 2016
Expert consensus on the clinical application of GnRH-a in obstetrics and gynecology Chinese Journal of Obstetrics and Gynecology, 2018
Agonist-induced internalization and downregulation of gonadotropin-releasing hormone receptors. Am J Physiol Cell Physiol, 2009
Triptorelin versus Leuprolide: A Comparative Analysis in Prostate Cancer. Urology, 2015
FAQ
- What is the use of triptorelin acetate 3.75 mg?
Triptorelin injection is used to treat certain types of prostate cancer. It is also used to treat central precocious puberty (CPP; a condition causing children to enter puberty too soon, resulting in faster than normal bone growth and development of sexual characteristics).
- What is triptorelin acetate injection 3.75 mg used for in IVF?
Decapeptyl (triptorelin) is a gonadotropin-releasing hormone (GnRH) agonist analogue drug. It is used in assisted reproduction treatments to control ovarian function in in vitro fertilization (IVF) cycles.
Hot Tags: triptorelin acetate 3.75 mg, China triptorelin acetate 3.75 mg manufacturers, suppliers