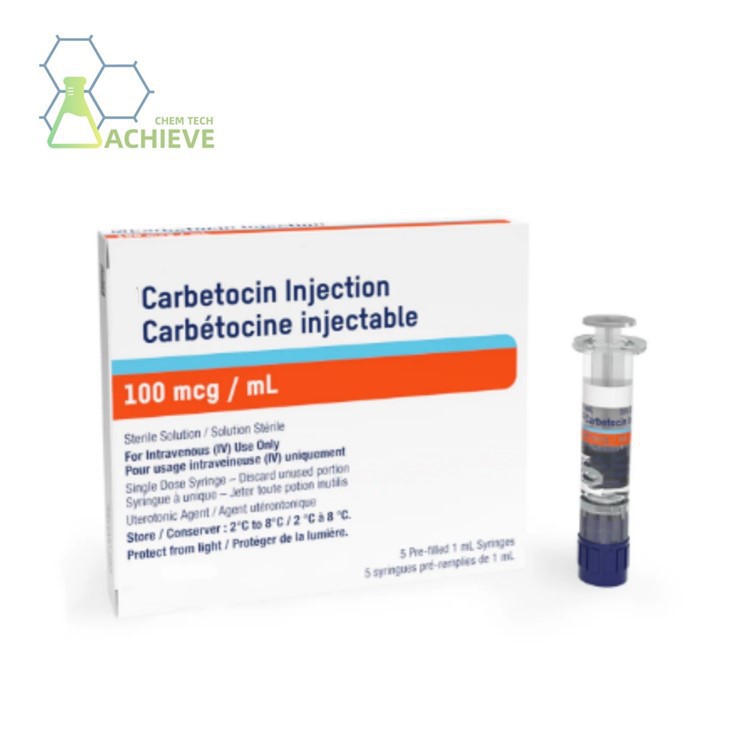
Carbetocin injection 100 mcg/ml is a long‑acting synthetic oxytocin injection specifically designed for the prevention of postpartum hemorrhage following cesarean section. Compared with conventional di-sipidin, it has a longer half‑life, more stable and controllable uterine contractions, minimal impact on hemodynamics, and an improved safety profile. With precise dosage and convenient administration, it is suitable for single intravenous bolus or intramuscular injection after delivery of the fetus under epidural or spinal anesthesia for cesarean section.
Products Description






Carbetocin\Carbetocin Acetate COA
 |
||
| Certificate of Analysis | ||
| Compound name | Carbetocin\Carbetocin Acetate | |
| Grade | Pharmaceutical grade | |
| CAS No. | 37025-55-1 | |
| Quantity | 36g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090056 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.54% |
| Loss on drying | ≤1.0% | 0.42% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.98% |
| Single impurity | <0.8% | 0.52% |
| Total microbial count | ≤750cfu/g | 95 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 500ppm |
| Storage |
Store in a sealed, dark, and dry place below -20°C |
|
|
|
||
|
|
||
| Chemical Formula | C45H69N11O12S |
| Exact Mass | 987.48 |
| Molecular Weight | 988.17 |
| m/z | 987.48(100.0%), 988.49(48.7%), 989.49(11.6%), 989.48(4.5%), 988.48(4.1%), 989.49(2.5%), 990.48(2.2%), 989.49(2.0%), 990.49(1.2%) |
| Elemental Analysis | C,54.70; H,7.04; N,15.59; O,19.43; S,3.24 |

Prevention of Uterine Atony Following Cesarean Section
1.1 Core Indication
The primary clinical indication of carbetocin injection 100 mcg/ml is the prevention of uterine atony and subsequent reduction in the risk of postpartum hemorrhage following elective cesarean section.Cesarean section is a common obstetric intervention. According to the World Health Organization, its global incidence is approximately 15%, with substantial variation across countries and regions, reaching up to 52% in some nations. Due to uterine myometrial trauma during surgery, inhibitory effects of anesthetic agents on uterine contractility, and individual maternal differences, the incidence of uterine atony after cesarean section is significantly higher than that after vaginal delivery. Uterine atony is the leading cause of postpartum hemorrhage, which in severe cases can lead to maternal hemorrhagic shock, disseminated intravascular coagulation, and even death. Therefore, timely prevention of uterine atony in the postoperative period is critical.
1.2 Clinical Advantages
Compared with conventional di-sipidin, the product demonstrates distinct advantages in preventing post‑cesarean uterine atony, particularly in duration of action, ease of administration, and stability of clinical effect, as detailed below:
First, it has a significantly longer duration of action and superior long‑acting properties. Conventional di-sipidin has a short half‑life of only 4–10 minutes, with limited sustained uterine contractile effect. It typically requires continuous intravenous infusion to maintain therapeutic effect for postpartum hemorrhage prophylaxis, which increases nursing workload and may cause excessive or insufficient uterine contractions due to inappropriate infusion rates.

As a long‑acting di-sipidin analog, carbetocin exhibits biphasic elimination after intravenous administration, with a median terminal elimination half‑life of up to 33 minutes; the half‑life is even longer after intramuscular injection, with some studies showing sustained efficacy for several hours.
Continuous intravenous infusion is unnecessary, providing stable and prolonged uterine contraction and avoiding the need for repeated dosing or continuous infusion required for conventional di-sipidin.
Second, it is convenient to administer and simplifies clinical procedures. For postpartum hemorrhage prophylaxis after cesarean section, conventional di-sipidin requires an immediate intravenous bolus followed by continuous infusion for 4–6 hours, with strict rate control and considerable consumption of nursing resources. By contrast, carbetocin injection 100 mcg/ml only requires slow intravenous injection after fetal delivery, before or after placental delivery, with no need for subsequent continuous infusion. The simple administration regimen significantly reduces nursing workload, especially in busy obstetric settings, and lowers risks associated with improper infusion practices.

References
World Health Organization (WHO). WHO-PQ RECOMMENDED SUMMARY OF PRODUCT CHARACTERISTICS (RH 095) [R]. Undated.
Instituto de Salud Pública de Chile. FOLLETO DE INFORMACIÓN AL PROFESIONAL DURATOCIN SOLUCIÓN INYECTABLE 100 mcg/1 mL [R].
Adjunctive Management of Uterine Atonic Hemorrhage

Application Scenarios and Core Value
In addition to prophylaxis of post‑cesarean uterine atony, the product may be used as an adjunctive agent in combination with other uterotonics (e.g., conventional oxytocin, ergometrine, prostaglandins) for the emergency treatment of refractory postpartum hemorrhage.Postpartum hemorrhage is the leading cause of maternal mortality worldwide, affecting over 14 million women annually and resulting in 70,000 maternal deaths. Uterine atonic hemorrhage accounts for 70%–80% of postpartum hemorrhage cases. This condition is life‑threatening and requires rapid combination therapy with multiple uterotonics to enhance uterine contraction, control bleeding, and save the mother's life.
References
World Health Organization (WHO). WHO-PQ RECOMMENDED SUMMARY OF PRODUCT CHARACTERISTICS (RH 095) [R].
National Center for Biotechnology Information. Web annex 7 Choice of uterotonic agents [R]. Undated.
Application in Pharmacological Research Models of Uterine Smooth Muscle
The product serves as a key tool drug for pharmacological research on uterine smooth muscle. Relying on its long‑acting and stable agonistic effect on oxytocin receptors, it is widely used in in vitro, cellular and in vivo models to precisely elucidate uterine contraction mechanisms and drug effects.
In isolated uterine smooth muscle strip models, this preparation is commonly employed in organ bath experiments. Human or animal uterine muscle strips are placed in physiological solution, with cumulative administration of carbetocin injection 100 mcg/ml at concentrations ranging from 10⁻¹⁰ to 10⁻⁵ M. Its long‑acting activity stably sustains contractile responses for several hours, avoiding repeated dosing, making it suitable for studies on receptor desensitization, signaling pathways and synergistic effects of uterotonic agents.


In uterine smooth muscle cell models, it is used to detect intracellular calcium signals, myosin light chain phosphorylation and oxytocin receptor (OTR) activation pathways, confirming its mechanism of inducing contraction via the Gq‑PLC‑IP3‑Ca²⁺ pathway. It is also applied in screening OTR antagonists and evaluating the potency of uterotonic drugs, serving as a standard tool in pharmaceutical research and development.
In in vivo animal models (rats, guinea pigs, horses), single administration allows monitoring of uterine contraction dynamics and assessment of postpartum uterine involution and hemorrhage prevention efficacy. Its long‑acting property reduces dosing frequency and improves experimental stability.
References
American Society of Anesthesiologists. In Vitro Comparative Effect of Carbetocin and di-sipidin in Pregnant Human Myometrium[J]. Anesthesiology, 2026, 124(2):378‑387.
PubMed. Ex vivo influence of carbetocin on equine myometrial muscles and comparison with di-sipidin[J]. 2012, 76(3):245‑251.
1. API Synthetic Process
Carbetocin active pharmaceutical ingredient (API) is mainly manufactured via solid‑phase peptide synthesis, the mainstream method for peptide drugs due to its operational simplicity, high yield, and ease of purity control. Key steps include: using an amino resin as a solid support, deprotection, sequential coupling of Fmoc‑protected amino acids, removal of cysteine side‑chain protecting groups followed by attachment of bromobutyric acid, solid‑phase cyclization and cleavage to yield crude peptide, and final purification by reversed‑phase high‑performance liquid chromatography (RP‑HPLC) to obtain high‑purity API. Specific coupling and cyclization reagents are used during synthesis, with strict control of reaction conditions to ensure accurate amino acid sequence and minimize impurity formation.
2. Pharmaceutical Preparation Process
The clinically used carbetocin dosage form is injection, primarily at a strength of 1 mL:100 μg. The manufacturing process strictly adheres to aseptic processing standards. The formulation consists of carbetocin API, excipients, and water for injection. Excipients include mannitol, methionine, succinic acid, sodium hydroxide, etc., to adjust pH and enhance stability. During production, excipients are dissolved in water for injection, pH is adjusted to 3.0–5.5, purified carbetocin API is added and dissolved, followed by filtration, filling and sealing, and terminal sterilization to produce a clear, colorless solution. Filling accuracy and sterilization parameters are controlled to prevent microbial contamination and ensure compliance with injectable quality standards. The product is commercially manufactured by multiple domestic enterprises, with production sites located in Hangzhou, Zhongshan, Shenzhen, and other cities.
3. Quality Control Standards
Stringent quality control standards apply throughout the manufacturing of carbetocin, covering API, finished products, and storage and distribution.API testing includes appearance, identification, related substances, assay, water content, residual solvents, and microbial limits. Assay is performed by HPLC; nine known impurities are controlled to ensure acceptable purity.Finished injection testing includes description, pH, sterility, bacterial endotoxins, assay, and related substances. Sterility and bacterial endotoxin tests are critical for safety and must fully comply with pharmacopoeial and registration requirements.For storage, API should be kept light‑protected, sealed, and stored at −20°C. Finished injections must be stored under specified conditions. Once opened, API should be used in a single batch to avoid quality degradation due to moisture absorption.
References
National Institutes for Food and Drug Control. Instructions for National Drug Reference Substance: Carbetocin [R].
Journal of China Pharmaceutical University. Development and validation of a method for related substances testing in carbetocin injection [J].
World Health Organization (WHO). Carbetocin 100 mcg/mL solution for injection (Ferring International Center SA), RH095 [R]. November 2022.
Core Contraindications
Contraindicated during pregnancy and prior to delivery of the fetus.
Contraindicated in patients with hypersensitivity to carbetocin or di-sipidin.
Contraindicated in pediatric patients.
References
Carbetocin Injection Package Insert (Hainan Huanglong Pharmaceutical, 2023 edition); EMA Product Information: Pabal®; China Medical Information Query Platform; FDA Prescribing Information for Carbetocin.
FAQ
Is carbetocin given IV or IM?
+
-
DURATOCIN (carbetocin injection) is a long-acting synthetic octapeptide analogue of oxytocin with agonist properties. It can be administered intravenously as a single dose immediately following delivery by caesarean section under epidural or spinal anaesthesia, to prevent uterine atony and postpartum haemorrhage.
Why is carbetocin not used in PPH?
-
In contrast, heat-stable carbetocin is indicated only for PPH prevention. The inappropriate use of heat-stable carbetocin, similar to other existing uterotonics, can endanger the lives of women and babies. Education and monitoring, including pharmacovigilance, are paramount.
Hot Tags: carbetocin injection 100 mcg/ml, China carbetocin injection 100 mcg/ml manufacturers, suppliers, salcatonin injection, intranasal desmopressin acetate, best glp 1 peptide, best hcg supplement, glp1 shot, etelcalcetide structure