SLU-PP-332 Injection is an experimental drug candidate compound designed to simulate the effects of exercise. This substance works by targeting specific estrogen-related receptors, ERRα, which is a key protein regulating energy metabolism within cells. The core mechanism involves activating and enhancing the function of mitochondria, promoting the production of more energy carrier ATP by cells, thereby inducing physiological adaptations such as increased physical performance, accelerated metabolism, and enhanced muscle endurance similar to those after high-intensity endurance training. It is thus vividly referred to as an "exercise simulator" or "exercise pill". Currently, this compound is still in the preclinical research stage and is mainly used for scientific research to explore its potential for treating muscle atrophy, metabolic diseases (such as obesity and diabetes), and heart failure, which are related to metabolic and muscle function decline. It should be emphasized that it is not an approved drug, and its long-term safety and actual human efficacy still need to be verified through rigorous clinical trials in the future.
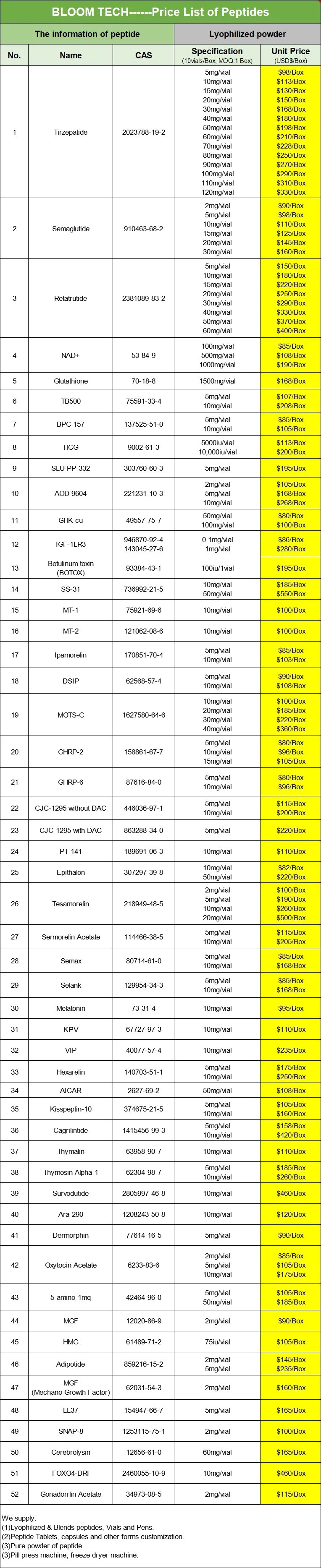
Our products form






SLU-PP-332 COA



SLU-PP-332 Injection is an experimental compound that mimics the metabolic effects of exercise by activating estrogen-related receptors (ERRs). Its core functions include increasing energy expenditure, promoting fatty acid oxidation, and optimizing mitochondrial function. The following are the detailed instructions regarding its usage:
Applicable Scenarios and Target Population
The potential application scenarios of SLU-PP-332 Injection include:
Metabolic disease treatment
For patients with metabolic syndrome such as obesity and insulin resistance, reduce fat accumulation and improve energy metabolism through non-exercise-dependent methods.
Muscle function assistance
Provide auxiliary treatment for patients with muscle atrophy caused by aging, cancer, or prolonged bed rest, inhibiting muscle breakdown and promoting synthesis.
Improvement of exercise endurance
Help athletes or rehabilitation patients enhance muscle endurance and optimize athletic performance.
Usage and Dosage
Dosage
In animal experiments, the common dosage is 10 mg/kg body weight. For example, in a mouse experiment where each individual weighs 20g, the volume of the solution for administration is 100μL, and the working solution concentration needs to be prepared as 2 mg/mL (the stock solution concentration is 40 mg/mL).
The human dosage has not been determined yet, and the safe range needs to be determined through subsequent clinical trials.
Administration method
Intraperitoneal injection: For compounds with high powder solubility, it is recommended to use salt-forming compounds or suspensions. For example, when administering a large dose, a suspension can be used for intraperitoneal injection.
Gavage administration: When the dose is large, it is recommended to prepare a uniform suspension with 0.5% carboxymethyl cellulose sodium (CMC-Na) for administration.
Other methods: According to experimental needs, intravenous injection or subcutaneous injection can also be explored, but safety and efficacy need to be verified.
Solvent selection and preparation
Base solvent: DMSO (dimethyl sulfoxide) is a commonly used solvent, but attention should be paid to the concentration limit. For normal mice, the DMSO concentration is recommended to be below 10%, and for nude mice or weak mice, it is recommended to be below 2%.
Composite solvent: To improve solubility and stability, a composite solvent formula can be used, such as 10% DMSO + 40% PEG300 + 5% Tween-80 + 45% normal saline/PBS/distilled water. The solvents should be added in sequence, fully dissolved before adding the next component.
Preparation steps: Taking a 10 mg/kg dosage as an example, if each animal weighs 20g and the administration volume is 100μL, 50μL of DMSO should be dissolved to prepare the compound, then 300μL of PEG300 should be added to mix evenly, followed by 50μL of Tween-80 to mix evenly, and finally 600μL of normal saline/PBS/distilled water should be added to make up the volume.
Operational Precautions

Solubility optimization
If the solubility of the compound is low, it can be assisted in dissolution by heating to 37°C or ultrasonic oscillation.
Avoid repeated freezing and thawing. The prepared solution should be aliquoted and stored separately, and used within the specified time (such as -80°C for 6 months, -20°C for 1 month).
Dosage adjustment
For large packaging compounds (such as 10 mg or more), it is recommended to batch weigh and prepare reserve solutions to reduce errors.
When preparing for the first time, a small amount of the compound should be tested for the solubility of the formula, and then a large amount should be prepared.


Animal experiment design
Considering the loss during the experiment, it is recommended to prepare the dosage for one animal more.
Adjust the solvent formula according to the animal species and administration method, for example, the DMSO concentration for nude mice should be reduced.
Safety and Monitoring

Side effect observation
In animal experiments, closely observe behavioral changes, food intake, body weight, and organ indices, etc.
Long-term use needs to assess potential effects on the reproductive system, liver, and kidneys.
Clinical translation challenges
SLU-PP-332 is currently still in preclinical research, and its safety and efficacy need to be further verified through human trials.
Attention should be paid to the risk of off-target effects, especially the association between ERRs and the reproductive system.

Sterility and Heat Sources
SLU-PP-332 Injection, as an experimental compound, aims to simulate the metabolic effects produced by exercise by activating estrogen-related receptors (ERRα, ERRβ, ERRγ). Its preclinical research has demonstrated the potential to improve metabolic syndrome, obesity, and muscle function. During the drug development process, aseptic and heat source control are the core aspects for ensuring the safety of the medication. The following analysis is conducted from three aspects: aseptic assurance, heat source control, potential risks, and improvement directions.
Sterility Assurance: Multiple Steps Work Together to Ensure the Sterility of the Preparation
The sterility of SLU-PP-3PP-332 Injection is achieved through strict production processes and quality control. At the raw material stage, the compound needs to be purified by high-performance liquid chromatography (HPLC) to a purity of over 98%, and aseptic crystallization technology is used to prevent microbial contamination. In terms of the production environment, the preparation workshop must comply with GMP standards, maintaining cleanliness through laminar flow purification systems, and key operations (such as filling and sealing) are completed in a hundred-grade clean area to minimize the risk of microbial contamination.
The choice of packaging materials is also crucial. Containers with good sealing properties, such as aluminum foil bags or glass bottles, can effectively prevent the invasion of external microorganisms. At the same time, the packaging itself must undergo sterilization verification (such as ethylene oxide sterilization or irradiation sterilization) to ensure sterility. Regarding storage conditions, powder preparations are recommended to be stored at -20℃ to maintain stability, while solutions prepared with solvents need to be stored at -80℃ for a short period or at -20℃ for a long period to avoid microbial growth caused by repeated freezing and thawing.
Heat Source Control: Dual measures of strict testing and process optimization
The heat source (endotoxin) is a component of the cell wall of Gram-negative bacteria. Injecting it into the human body may cause severe reactions such as fever and shock. The control of the heat source in SLU-PP-332 Injection should be carried out from three aspects: raw materials, production process, and product testing.

Raw material control
The raw medicinal substances need to be tested for endotoxin content using the Limulus Amoebocyte Lysate (LAL) method to ensure compliance with the "less than 0.1 EU per 1mg" standard stipulated in the "Chinese Pharmacopoeia".

Production process optimization
During the production process, the use of excipients containing heat sources (such as certain surfactants) should be avoided. At the same time, potential heat sources can be removed through ultrafiltration, distillation, etc. For example, filtration with a 0.22μm filter membrane can effectively retain bacteria and large molecular endotoxins.

Finished product testing
The finished products need to undergo endotoxin quantitative testing to ensure that the endotoxin content in each injection is less than 5EU/kg (the maximum safe dose for humans). For high-risk preparations (such as intravenous injections), more stringent testing standards (such as less than 0.5EU/mg) should be adopted.
Potential Risks and Improvement Directions
Although SLU-PP-332 Injection did not report any serious adverse reactions in preclinical studies, its aseptic and heat source control still require attention to the following risks:
If biological catalysts (such as enzymes) or animal-derived excipients are used in the synthesis process of the raw materials, exogenous heat sources may be introduced. Improvement directions include using recombinant enzymes instead of animal enzymes and strengthening the audit of raw material suppliers.
Improper control of temperature, humidity, and pressure difference in the clean area may lead to microbial proliferation. Real-time monitoring systems (such as particle counters, temperature and humidity sensors) should be used to ensure stable environmental parameters.
Loose packaging seals may allow microbial invasion. Packaging integrity should be verified through packaging sealing tests (such as vacuum attenuation method, color water method).
In addition, for the special properties of SLU-PP-332 Injection (such as the use of DMSO solvent), the interference of DMSO on heat source detection needs to be evaluated, and the detection method (such as dilution method or dedicated reagent kit) should be optimized.
FAQ
1. What is SLU-PP-332 injection?
It is an experimental compound injection solution in the strict preclinical research stage. Its core function is to activate the ERRα protein within cells and simulate some of the metabolic enhancement effects brought about by exercise. Currently, it is only used for scientific research and there are no approved human injection products on the market.
2. Can it safely "simulate the effects of exercise"?
Absolutely not. The existing data is based only on cell and animal experiments and has not proven its safety and effectiveness for humans. Self-injection cannot replicate the complex benefits of exercise on the entire body system, and it may cause serious and irreversible health damage.
3. Is there a possibility that SLU-PP-332 injection could become a prescription drug?
There is a possibility, but the road is long and uncertain. Its prototype compound is only used as an important "tool molecule" to verify the scientific value of the ERRα target. If it is to be developed into a drug, it needs to undergo comprehensive chemical structure optimization and lengthy, strict clinical trials over several years to prove its safety and effectiveness.
Hot Tags: slu-pp-332 injection, China slu-pp-332 injection manufacturers, suppliers, AOD 9604 Injection, AOD 9604 Spray, bodybuilding peptide, Semaglutide Capsule, Semaglutide Spray, SLU PP 332 250mcg












