Orforglipron Peptide is a novel oral GLP-1 receptor agonist peptide drug with a unique structure and superior efficacy. This peptide drug features flexible conformational characteristics, with its key torsion angles capable of dynamic adjustment, enabling it to enter the GLP-1 receptor binding pocket via an induced-fit mechanism. It enhances binding stability through hydrophobic interactions and hydrogen bonds, preferentially activates the Gs protein signaling pathway, and reduces receptor desensitization.
Our Products Form




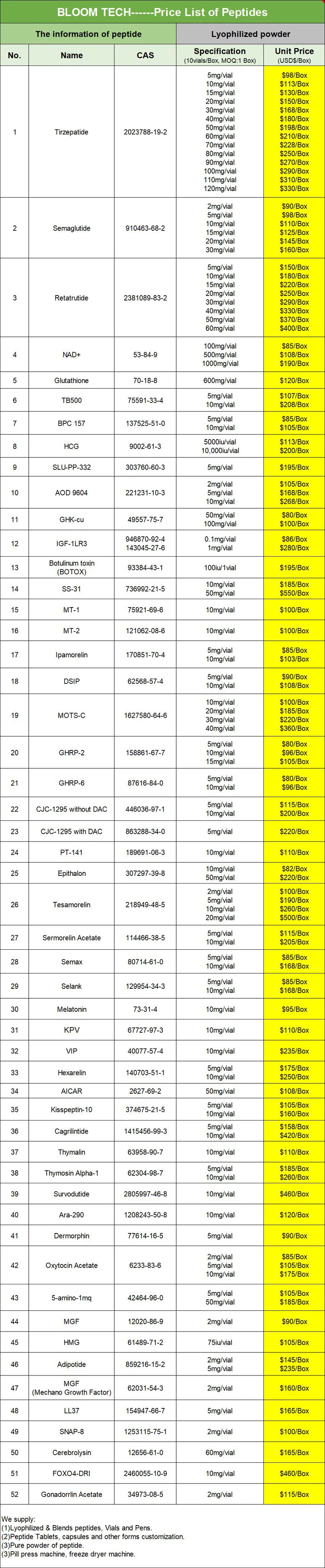


Orforglipron COA



Application in the Treatment of Stress Urinary Incontinence (SUI)
Pathogenesis of Stress Urinary Incontinence (SUI)
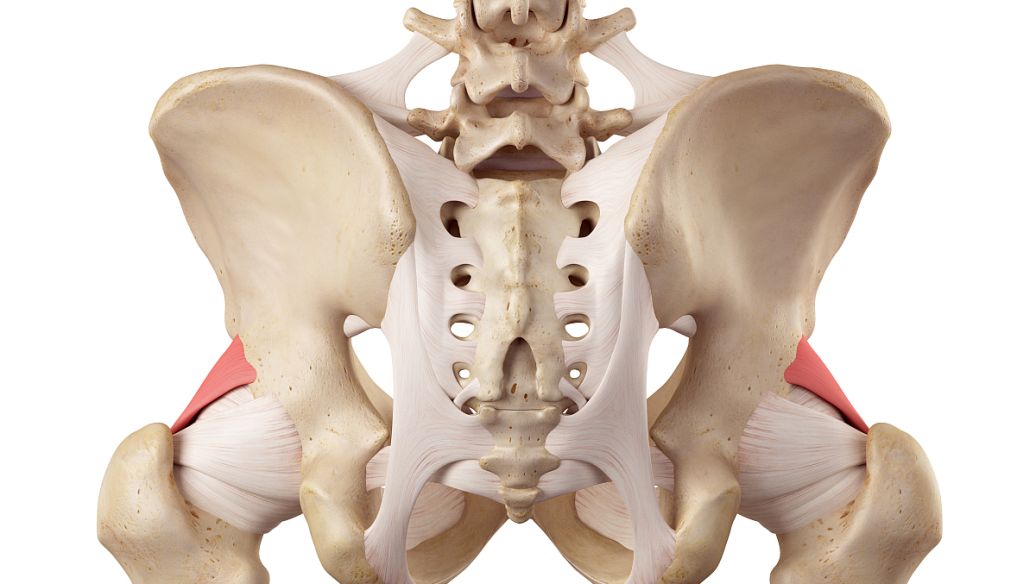
The pathogenesis of SUI is complex, with the core abnormality lying in dysfunction of the urinary control system, mainly involving multiple aspects including the pelvic floor muscles, urethral sphincter, urethral support structures, and neural regulation. Among these, obesity is one of the key risk factors inducing or exacerbating SUI. Clinical data show that women with a higher body mass index (BMI) have a significantly increased risk of SUI. Chronic obesity leads to persistently elevated abdominal pressure, compressing the pelvic floor muscles, causing muscle relaxation or nerve injury, thereby reducing the closure capacity of the urethral sphincter, and ultimately resulting in involuntary urine leakage when abdominal pressure increases.
In addition, pelvic floor muscle injury in women after childbirth, urethral mucosal atrophy and loosening of periurethral connective tissue caused by decreased estrogen levels post-menopause, as well as age-related degenerative changes in pelvic floor muscle function, are also major predisposing factors for SUI.
From the perspective of physiological regulation, normal urinary control function of the bladder and urethra relies on neuromuscular coordination. GLP-1 receptors are expressed in the bladder, urethra, and pelvic floor muscle tissues, providing an important molecular basis for Orforglipron Peptide to intervene in SUI-by activating GLP-1 receptors, it can modulate nerve conduction and muscle contraction, improve the stability of the urinary control system, and thus alleviate SUI symptoms.
Mechanism of Action in SUI Treatment
The mechanism of action of Orforglipron in SUI treatment is complex. Its core effect is to repair urinary control system function and relieve SUI symptoms through synergistic effects on three dimensions: weight loss, neural regulation, and muscle function improvement by activating GLP-1 receptors. These mechanisms are interrelated and mutually reinforcing, as detailed below:
(1) Reducing Pelvic Floor Muscle Burden at the Source
As mentioned above, obesity is a major trigger of SUI. Long-term high BMI leads to sustained high abdominal pressure, compressing pelvic floor muscles, causing muscle relaxation and nerve damage, and further weakening urethral sphincter closure. By activating GLP-1 receptors, Orforglipron specifically acts on the hypothalamic appetite center to suppress appetite and reduce food intake, while delaying gastric emptying and enhancing satiety, thereby achieving dose-dependent weight loss.
In addition, it regulates lipid metabolism and reduces fat accumulation, especially abdominal fat, further lowering abdominal pressure. Clinical data show that participants in the Orforglipron 36 mg group achieved an average weight reduction of 8.9 kg (9.2%), significantly superior to 5.0 kg (5.3%) in the oral semaglutide 14 mg group, with marked and durable weight loss.
Effective weight reduction fundamentally relieves abdominal compression on pelvic floor tissues, alleviates pelvic floor muscle relaxation, restores normal urethral sphincter closure, and reduces urine leakage under increased abdominal pressure. This represents one of the core mechanisms for SUI treatment, particularly suitable for SUI patients with obesity or overweight.
(2) Improving Synergistic Function of the Bladder and Urethra
GLP-1 receptors are expressed not only in pancreatic islets and the gastrointestinal tract but also widely in nerve endings of the bladder, urethra, and pelvic floor muscles, participating in the neural regulation of detrusor contraction and urethral sphincter closure. By selectively activating GLP-1 receptors, Orforglipron modulates conduction in spinal and pelvic nerves, inhibits overactive detrusor contraction, enhances bladder storage capacity, and promotes urethral sphincter contraction to increase urethral closure pressure and reduce involuntary urine leakage.
Furthermore, Orforglipron Peptide regulates pelvic nerve sensitivity, reduces abnormal nerve signal transmission caused by pelvic floor muscle relaxation and nerve injury, relieves concomitant symptoms such as urinary frequency and urgency, and further improves patients' urinary control. This neural regulatory effect not only alleviates SUI symptoms but also improves lower urinary tract function, enhancing therapeutic comprehensiveness.
(3) Strengthening Pelvic Floor Muscle and Urethral Sphincter Function
Pelvic floor muscle relaxation and reduced urethral sphincter function are core pathological changes in SUI. Orforglipron improves these functions through multiple pathways. On the one hand, its weight-lowering effect reduces the burden on pelvic floor muscles and creates conditions for muscle repair. On the other hand, by activating GLP-1 receptors, Orforglipron promotes the proliferation and differentiation of pelvic floor muscle cells, enhances muscle contractility, and inhibits degenerative changes to slow muscle relaxation.
In addition, GLP-1 receptor activation promotes urethral mucosal repair and hyperplasia, increasing mucosal thickness and elasticity to improve urethral sealing and reduce urine leakage. Preclinical studies indicate that Orforglipron treatment significantly increases the amplitude and duration of pelvic floor muscle contraction and strengthens urethral sphincter closure, providing direct histological support for SUI treatment.
Advantages and Limitations of Orforglipron in SUI Treatment
(1) Core Application Advantages
Compared with existing SUI treatment regimens, Orforglipron shows distinct advantages in four main aspects:
First, significant integrated therapeutic benefit. Orforglipron combines weight loss and urinary control effects, simultaneously addressing the core risk factor (obesity) and clinical symptoms of SUI. It is especially suitable for SUI patients with obesity or overweight, achieving the dual goals of "weight loss + urinary control" and reducing SUI recurrence at the source. Its efficacy is more comprehensive and durable than traditional monotherapies.
Second, convenient administration and high compliance. Administered orally once daily without dietary or water intake restrictions, it requires no injection or refrigeration, aligning with daily living habits.
It effectively solves the poor compliance associated with conventional treatments (e.g., pelvic floor muscle training, injectable drugs) and is suitable for long-term use, particularly for elderly or mobility-impaired SUI patients.
Third, favorable safety and high tolerability. Adverse events are mostly mild-to-moderate gastrointestinal reactions that resolve spontaneously with prolonged treatment, with no severe adverse effects. It avoids risks related to estrogen medications and surgical interventions, expanding the eligible population to include SUI patients with mild cardiovascular diseases or diabetes (used under medical supervision).
Fourth, high target selectivity and clear efficacy. By targeting GLP-1 receptor activation, it acts synergistically across neural, muscular, and metabolic dimensions to precisely improve urinary control. Preliminary clinical observations show a symptom relief rate of over 60% in patients with mild-to-moderate SUI, with efficacy dose-dependent, allowing individualized dosing based on disease severity.
(2) Current Limitations
Despite promising potential in SUI treatment, Orforglipron Peptide still has several limitations:
First, clinical trials are incomplete, requiring further validation of efficacy and safety. Phase 3 clinical trials for Orforglipron in SUI are ongoing, with no finalized results. Key data including long-term efficacy, optimal therapeutic dose, and recurrence rate remain to be confirmed, limiting broad clinical application.
Second, certain adverse reactions and contraindications exist. Although adverse events are mostly mild-to-moderate, some patients may experience nausea, diarrhea, and other discomforts. Orforglipron is contraindicated in patients with hypersensitivity to the drug or its ingredients, severe gastrointestinal diseases, or severe hepatic and renal insufficiency, restricting use in some special populations.
Third, relatively high drug cost. As a novel oral GLP-1 receptor agonist, Orforglipron involves high R&D costs. If approved, its price may exceed that of traditional SUI drugs, potentially increasing patient economic burden and affecting widespread adoption.

With advancing clinical research, Orforglipron holds broad prospects in SUI management. Future breakthroughs may focus on the following directions to optimize the SUI treatment system:
(1) Combination Therapy to Enhance Efficacy
Future research may explore combination regimens of Orforglipron with existing SUI treatments, such as pelvic floor muscle training and biofeedback therapy. Leveraging Orforglipron's weight loss and neural regulation effects combined with muscle function improvement from pelvic floor training can achieve synergistic enhancement, further improving outcomes in moderate-to-severe SUI patients.
Combination with surgery is another direction: preoperative Orforglipron can reduce weight and improve pelvic floor function to lower surgical difficulty and postoperative recurrence risk, while postoperative use promotes recovery and enhances surgical efficacy.
(2) Drug Design Optimization to Reduce Treatment Costs
With advances in R&D technology, future optimization of Orforglipron's molecular structure can improve bioavailability, reduce effective dosage and adverse reactions, while lowering R&D and production costs to make it more affordable and accessible. Development of long-acting formulations, such as once-weekly oral preparations, will further simplify administration and improve patient compliance.
(3) In-Depth Mechanistic Research to Expand Therapeutic Indications
Further studies are needed to elucidate the specific molecular pathways of Orforglipron Peptide in neural regulation, muscle repair, and metabolic modulation during SUI treatment, providing a theoretical basis for drug optimization and upgrading.
Meanwhile, exploration of GLP-1 receptor agonists in other forms of urinary incontinence (e.g., urge urinary incontinence) will expand their clinical scope and offer new strategies for the treatment of urinary system diseases.

I. Driven by Unmet Clinical Needs
The discovery of Orforglipron stems from the urgent global demand for convenient and highly effective metabolic drugs. With the rising incidence of type 2 diabetes and obesity, traditional GLP‑1 receptor agonists are mostly peptide-based injectable formulations, which suffer from drawbacks such as inconvenient administration and poor patient compliance. Therefore, the development of novel, oral, and potent GLP‑1 receptor agonists has become a major focus in the pharmaceutical industry. Chugai Pharmaceutical (Japan) took the lead in launching relevant research, focusing on non-peptide small-molecule design, aiming to overcome the limitations of conventional peptide drugs.
II. Breakthrough Research and Development of Small-Molecule Structure
Around 2017, the research team at Chugai Pharmaceutical successfully identified a small-molecule compound capable of specifically activating the GLP‑1 receptor, namely Orforglipron (research code: OWL833, later renamed LY3502970). Its core breakthrough lies in the non-peptide molecular structure, with a molecular weight of only 357.44 Da. It is resistant to degradation by gastrointestinal enzymes, shows promising potential for oral absorption, and exhibits high affinity and strong targeting to the GLP‑1 receptor. This solves the long-standing problems of low bioavailability and injectable administration associated with traditional peptide drugs.

III. Global R&D and Commercialization Licensing
In September 2018, Chugai Pharmaceutical signed an exclusive global license agreement with Eli Lilly and Company (USA), granting Lilly worldwide rights to the development and commercialization of Orforglipron, at which time the drug was about to enter Phase I clinical trials. Subsequently, Lilly led the advancement of clinical trials, gradually verifying its efficacy and safety in weight management and glycemic control. Phase II clinical results were released in 2023, followed by multiple positive Phase III clinical data in 2025. Marketing applications have now been submitted to more than 40 countries worldwide, marking a new stage in its clinical application.
FAQ
Is orforglipron a peptide?
+
-
Orforglipron, a small-molecule, nonpeptide oral glucagon-like peptide-1 (GLP-1) receptor agonist, is being investigated as a treatment for obesity.
Is orforglipron as effective as tirzepatide?
+
-
Which is better for weight loss? Talking purely about results, tirzepatide is more effective. But that doesn't mean it's best for you personally. At Voy, our clinicians recommend weight loss medications based on your individual health and medical history.
Hot Tags: orforglipron peptide, China orforglipron peptide manufacturers, suppliers, Best HCG Drops, CJC 1295 Cream, CJC 1295 Injection, CJC 1295 Tablets, IGF 1 LR3 Spray, MT2 Injection





















